Rescue of Notch signaling in cells incapable of GDP-L-fucose synthesis by gap junction transfer of GDP-L-fucose in Drosophila
- PMID: 22949680
- PMCID: PMC3458340
- DOI: 10.1073/pnas.1202369109
Rescue of Notch signaling in cells incapable of GDP-L-fucose synthesis by gap junction transfer of GDP-L-fucose in Drosophila
Abstract
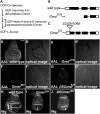
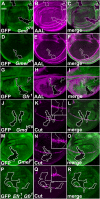
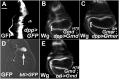
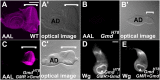
Notch (N) is a transmembrane receptor that mediates cell-cell interactions to determine many cell-fate decisions. N contains EGF-like repeats, many of which have an O-fucose glycan modification that regulates N-ligand binding. This modification requires GDP-L-fucose as a donor of fucose. The GDP-L-fucose biosynthetic pathways are well understood, including the de novo pathway, which depends on GDP-mannose 4,6 dehydratase (Gmd) and GDP-4-keto-6-deoxy-D-mannose 3,5-epimerase/4-reductase (Gmer). However, the potential for intercellularly supplied GDP-L-fucose and the molecular basis of such transportation have not been explored in depth. To address these points, we studied the genetic effects of mutating Gmd and Gmer on fucose modifications in Drosophila. We found that these mutants functioned cell-nonautonomously, and that GDP-L-fucose was supplied intercellularly through gap junctions composed of Innexin-2. GDP-L-fucose was not supplied through body fluids from different isolated organs, indicating that the intercellular distribution of GDP-L-fucose is restricted within a given organ. Moreover, the gap junction-mediated supply of GDP-L-fucose was sufficient to support the fucosylation of N-glycans and the O-fucosylation of the N EGF-like repeats. Our results indicate that intercellular delivery is a metabolic pathway for nucleotide sugars in live animals under certain circumstances.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Biochemical characterization of GDP-L-fucose de novo synthesis pathway in fungus Mortierella alpina.Biochem Biophys Res Commun. 2010 Jan 22;391(4):1663-9. doi: 10.1016/j.bbrc.2009.12.116. Epub 2009 Dec 24. Biochem Biophys Res Commun. 2010. PMID: 20035716
-
Functional expression of Escherichia coli enzymes synthesizing GDP-L-fucose from inherent GDP-D-mannose in Saccharomyces cerevisiae.Glycobiology. 2000 Oct;10(10):1041-7. doi: 10.1093/glycob/10.10.1041. Glycobiology. 2000. PMID: 11030750
-
Two pathways for importing GDP-fucose into the endoplasmic reticulum lumen function redundantly in the O-fucosylation of Notch in Drosophila.J Biol Chem. 2010 Feb 5;285(6):4122-4129. doi: 10.1074/jbc.M109.016964. Epub 2009 Nov 30. J Biol Chem. 2010. PMID: 19948734 Free PMC article.
-
Metabolism and transportation pathways of GDP-fucose that are required for the O-fucosylation of Notch.Adv Exp Med Biol. 2012;727:37-46. doi: 10.1007/978-1-4614-0899-4_3. Adv Exp Med Biol. 2012. PMID: 22399337 Review.
-
The metabolism of 6-deoxyhexoses in bacterial and animal cells.Biochimie. 1998 Nov;80(11):923-31. doi: 10.1016/s0300-9084(00)88889-6. Biochimie. 1998. PMID: 9893952 Review.
Cited by
-
Molecular and morphological approach to study the innexin gap junctions in Rhynchosciara americana.Open Biol. 2021 Nov;11(11):210224. doi: 10.1098/rsob.210224. Epub 2021 Nov 10. Open Biol. 2021. PMID: 34753320 Free PMC article.
-
Dual Roles of O-Glucose Glycans Redundant with Monosaccharide O-Fucose on Notch in Notch Trafficking.J Biol Chem. 2016 Jun 24;291(26):13743-52. doi: 10.1074/jbc.M115.710483. Epub 2016 Apr 25. J Biol Chem. 2016. PMID: 27129198 Free PMC article.
-
Down-regulation of α-L-fucosidase 1 expression confers inferior survival for triple-negative breast cancer patients by modulating the glycosylation status of the tumor cell surface.Oncotarget. 2015 Aug 28;6(25):21283-300. doi: 10.18632/oncotarget.4238. Oncotarget. 2015. PMID: 26204487 Free PMC article.
-
Comparison of predicted epimerases and reductases of the Campylobacter jejuni D-altro- and L-gluco-heptose synthesis pathways.J Biol Chem. 2013 Jul 5;288(27):19569-80. doi: 10.1074/jbc.M113.468066. Epub 2013 May 20. J Biol Chem. 2013. PMID: 23689373 Free PMC article.
-
Two classes of gap junction channels mediate soma-germline interactions essential for germline proliferation and gametogenesis in Caenorhabditis elegans.Genetics. 2014 Nov;198(3):1127-53. doi: 10.1534/genetics.114.168815. Epub 2014 Sep 6. Genetics. 2014. PMID: 25195067 Free PMC article.
References
-
- Bishop JR, Schuksz M, Esko JD. Heparan sulphate proteoglycans fine-tune mammalian physiology. Nature. 2007;446:1030–1037. - PubMed
-
- Brückner K, Perez L, Clausen H, Cohen S. Glycosyltransferase activity of Fringe modulates Notch-Delta interactions. Nature. 2000;406:411–415. - PubMed
-
- Moloney DJ, et al. Fringe is a glycosyltransferase that modifies Notch. Nature. 2000;406:369–375. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

