An antitubulin agent BCFMT inhibits proliferation of cancer cells and induces cell death by inhibiting microtubule dynamics
- PMID: 22952952
- PMCID: PMC3432122
- DOI: 10.1371/journal.pone.0044311
An antitubulin agent BCFMT inhibits proliferation of cancer cells and induces cell death by inhibiting microtubule dynamics
Abstract
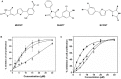
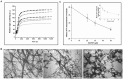
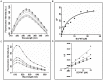
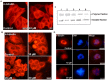
Using cell based screening assay, we identified a novel anti-tubulin agent (Z)-5-((5-(4-bromo-3-chlorophenyl)furan-2-yl)methylene)-2-thioxothiazolidin-4-one (BCFMT) that inhibited proliferation of human cervical carcinoma (HeLa) (IC(50), 7.2 ± 1.8 µM), human breast adenocarcinoma (MCF-7) (IC(50), 10.0 ± 0.5 µM), highly metastatic breast adenocarcinoma (MDA-MB-231) (IC(50), 6.0 ± 1 µM), cisplatin-resistant human ovarian carcinoma (A2780-cis) (IC(50), 5.8 ± 0.3 µM) and multi-drug resistant mouse mammary tumor (EMT6/AR1) (IC(50), 6.5 ± 1 µM) cells. Using several complimentary strategies, BCFMT was found to inhibit cancer cell proliferation at G2/M phase of the cell cycle apparently by targeting microtubules. In addition, BCFMT strongly suppressed the dynamics of individual microtubules in live MCF-7 cells. At its half maximal proliferation inhibitory concentration (10 µM), BCFMT reduced the rates of growing and shortening phases of microtubules in MCF-7 cells by 37 and 40%, respectively. Further, it increased the time microtubules spent in the pause (neither growing nor shortening detectably) state by 135% and reduced the dynamicity (dimer exchange per unit time) of microtubules by 70%. In vitro, BCFMT bound to tubulin with a dissociation constant of 8.3 ± 1.8 µM, inhibited tubulin assembly and suppressed GTPase activity of microtubules. BCFMT competitively inhibited the binding of BODIPY FL-vinblastine to tubulin with an inhibitory concentration (K(i)) of 5.2 ± 1.5 µM suggesting that it binds to tubulin at the vinblastine site. In cultured cells, BCFMT-treatment depolymerized interphase microtubules, perturbed the spindle organization and accumulated checkpoint proteins (BubR1 and Mad2) at the kinetochores. BCFMT-treated MCF-7 cells showed enhanced nuclear accumulation of p53 and its downstream p21, which consequently activated apoptosis in these cells. The results suggested that BCFMT inhibits proliferation of several types of cancer cells including drug resistance cells by suppressing microtubule dynamics and indicated that the compound may have chemotherapeutic potential.
Conflict of interest statement
Figures

References
-
- O'Connor R, Clynes M, Dowling P, O'Donovan N, O'Driscoll L (2007) Drug resistance in cancer-searching for mechanisms markers and therapeutic agents. Expert Opin Drug Metab Toxicol 3: 805–817. - PubMed
-
- Kavallaris M (2010) Microtubules and resistance to tubulin-binding agents. Nat Rev Cancer 10: 194–204. - PubMed
-
- Drews J (2000) Drug discovery: a historical perspective. Science 17: 1960–1964. - PubMed
-
- Balis FM (2002) Evolution of anticancer drug discovery and the role of cell-based screening. J Natl Cancer Inst 16: 78–79. - PubMed
-
- Sharma SV, Haber DA, Settleman J (2008) Cell line-based platforms to evaluate the therapeutic efficacy of candidate anticancer agents. Nat Rev Cancer 10: 241–253. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

