Brain-derived neurotrophic factor enhances calcium regulatory mechanisms in human airway smooth muscle
- PMID: 22952960
- PMCID: PMC3430656
- DOI: 10.1371/journal.pone.0044343
Brain-derived neurotrophic factor enhances calcium regulatory mechanisms in human airway smooth muscle
Abstract
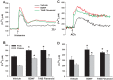
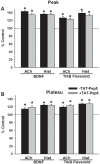
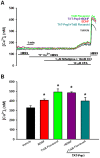
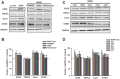
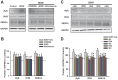
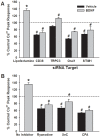
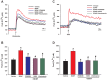
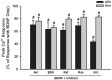
Neurotrophins (NTs), which play an integral role in neuronal development and function, have been found in non-neuronal tissue (including lung), but their role is still under investigation. Recent reports show that NTs such as brain-derived neurotrophic factor (BDNF) as well as NT receptors are expressed in human airway smooth muscle (ASM). However, their function is still under investigation. We hypothesized that NTs regulate ASM intracellular Ca(2+) ([Ca(2+)](i)) by altered expression of Ca(2+) regulatory proteins. Human ASM cells isolated from lung samples incidental to patient surgery were incubated for 24 h (overnight) in medium (control) or 1 nM BDNF in the presence vs. absence of inhibitors of signaling cascades (MAP kinases; PI3/Akt; NFκB). Measurement of [Ca(2+)](i) responses to acetylcholine (ACh) and histamine using the Ca(2+) indicator fluo-4 showed significantly greater responses following BDNF exposure: effects that were blunted by pathway inhibitors. Western analysis of whole cell lysates showed significantly higher expression of CD38, Orai1, STIM1, IP(3) and RyR receptors, and SERCA following BDNF exposure, effects inhibited by inhibitors of the above cascades. The functional significance of BDNF effects were verified by siRNA or pharmacological inhibition of proteins that were altered by this NT. Overall, these data demonstrate that NTs activate signaling pathways in human ASM that lead to enhanced [Ca(2+)](i) responses via increased regulatory protein expression, thus enhancing airway contractility.
Conflict of interest statement
Figures
References
-
- Friedman WJ, Greene LA (1999) Neurotrophin signaling via Trks and p75. Exp Cell Res 253: 131–142. - PubMed
-
- Kaplan DR, Miller FD (2000) Neurotrophin signal transduction in the nervous system. Curr Opin Neurobiol 10: 381–391. - PubMed
-
- Zweifel LS, Kuruvilla R, Ginty DD (2005) Functions and mechanisms of retrograde neurotrophin signalling. Nat Rev Neurosci 6: 615–625. - PubMed
-
- Chao MV (1994) The p75 neurotrophin receptor. J Neurobiol 25: 1373–1385. - PubMed
-
- Kalb R (2005) The protean actions of neurotrophins and their receptors on the life and death of neurons. Trends Neurosci 28: 5–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

