Interleukin-10 inhibits lipopolysaccharide-induced tumor necrosis factor-α translation through a SHIP1-dependent pathway
- PMID: 22955274
- PMCID: PMC3488072
- DOI: 10.1074/jbc.M112.348599
Interleukin-10 inhibits lipopolysaccharide-induced tumor necrosis factor-α translation through a SHIP1-dependent pathway
Abstract
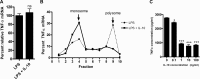
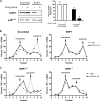
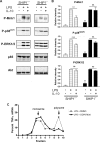
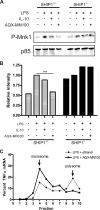
Production of the proinflammatory cytokine TNFα by activated macrophages is an important component of host defense. However, TNFα production must be tightly controlled to avoid pathological consequences. The anti-inflammatory cytokine IL-10 inhibits TNFα mRNA expression through activation of the STAT3 transcription factor pathway and subsequent expression of STAT3-dependent gene products. We hypothesized that IL-10 must also have more rapid mechanisms of action and show that IL-10 rapidly shifts existing TNFα mRNA from polyribosome-associated polysomes to monosomes. This translation suppression requires the presence of SHIP1 (SH2 domain-containing inositol 5'-phosphatase 1) and involves inhibition of Mnk1 (MAPK signal-integrating kinase 1). Furthermore, activating SHIP1 using a small-molecule agonist mimics the inhibitory effect of IL-10 on Mnk1 phosphorylation and TNFα translation. Our data support the existence of an alternative STAT3-independent pathway through SHIP1 for IL-10 to regulate TNFα translation during the anti-inflammatory response.
Figures
References
-
- Chow J. C., Young D. W., Golenbock D. T., Christ W. J., Gusovsky F. (1999) Toll-like receptor-4 mediates lipopolysaccharide-induced signal transduction. J. Biol. Chem. 274, 10689–10692 - PubMed
-
- Kruys V., Kemmer K., Shakhov A., Jongeneel V., Beutler B. (1992) Constitutive activity of the tumor necrosis factor promoter is canceled by the 3′-untranslated region in nonmacrophage cell lines; a trans-dominant factor overcomes this suppressive effect. Proc. Natl. Acad. Sci. U.S.A. 89, 673–677 - PMC - PubMed
-
- Buxadé M., Parra J. L., Rousseau S., Shpiro N., Marquez R., Morrice N., Bain J., Espel E., Proud C. G. (2005) The Mnks are novel components in the control of TNFα biosynthesis and phosphorylate and regulate hnRNP A1. Immunity 23, 177–189 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

