The extracellular matrix modulates fibroblast phenotype and function in the infarcted myocardium
- PMID: 22956156
- PMCID: PMC3535072
- DOI: 10.1007/s12265-012-9406-3
The extracellular matrix modulates fibroblast phenotype and function in the infarcted myocardium
Abstract
Cardiac fibroblasts are key cellular effectors of cardiac repair; their phenotype and function are modulated by interactions with extracellular matrix proteins. This review manuscript discusses the effects of the extracellular matrix on the inflammatory and reparative properties of fibroblasts in the infarcted myocardium. Early generation of matrix fragments in the infarct induces a pro-inflammatory and matrix-degrading fibroblast phenotype. Formation of a fibrin/fibronectin-rich provisional matrix serves as a conduit for migration of fibroblasts into the infarcted area. Induction of ED-A fibronectin and nonfibrillar collagens may contribute to myofibroblast transdifferentiation. Upregulation of matricellular proteins promotes transduction of growth factor and cytokine-mediated signals. As the scar matures, matrix cross-linking, clearance of matricellular proteins, and reduced growth factor signaling cause deactivation and apoptosis of reparative infarct fibroblasts. Understanding the effects of matrix components on infarct fibroblasts may guide the design of peptides that reproduce, or inhibit, specific matricellular functions, attenuating adverse remodeling.
Figures

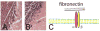
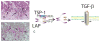
References
-
- Nag AC. Study of non-muscle cells of the adult mammalian heart: a fine structural analysis and distribution. Cytobios. 1980;28:41–61. - PubMed
-
- Baudino TA, Carver W, Giles W, Borg TK. Cardiac fibroblasts: friend or foe? Am J Physiol Heart Circ Physiol. 2006;291:H1015–1026. - PubMed
-
- Porter KE, Turner NA. Cardiac fibroblasts: at the heart of myocardial remodeling. Pharmacol Ther. 2009;123:255–278. - PubMed
-
- Camelliti P, Borg TK, Kohl P. Structural and functional characterisation of cardiac fibroblasts. Cardiovasc Res. 2005;65:40–51. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

