New developments in the treatment of actinic keratosis: focus on ingenol mebutate gel
- PMID: 22956883
- PMCID: PMC3430094
- DOI: 10.2147/CCID.S28905
New developments in the treatment of actinic keratosis: focus on ingenol mebutate gel
Abstract
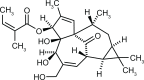
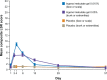
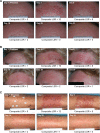
Actinic keratosis is a common disease in older, fair-skinned people, and is a consequence of cumulative ultraviolet exposure. It is part of a disease continuum in photodamaged skin that may lead to invasive squamous cell carcinoma. Treatment options frequently used include cryosurgery and topical pharmacologic agents, which are examples of lesion-directed and field-directed strategies. Ingenol mebutate gel was recently approved by the US Food and Drug Administration for topical treatment of actinic keratosis. While the mechanism of action of ingenol mebutate is not fully understood, in vitro and in vivo studies using tumor models indicate it has multiple mechanisms. Ingenol mebutate directly induces cell death by mitochondrial swelling and loss of cell membrane integrity preferentially in transformed keratinocytes. It promotes an inflammatory response characterized by infiltration of neutrophils and other immunocompetent cells that kills remaining tumor cells. The ability of ingenol mebutate to eliminate mutant p53 patches in ultraviolet-irradiated mouse skin suggests that it may have the potential to treat chronically ultraviolet-damaged skin. In human studies, ingenol mebutate achieved high clearance of actinic keratosis on the head and body after 2-3 consecutive daily treatments when measured by complete or partial clearance of lesions. Localized inflammatory skin responses were generally mild to moderate and resolved in less than a month.
Keywords: actinic keratosis; field therapy; ingenol mebutate gel; nonmelanoma skin cancer; protein kinase C delta.
Figures
References
-
- Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for actinic keratoses. Committee on Guidelines of Care. J Am Acad Dermatol. 1995;32(1):95–98. - PubMed
-
- Society for Investigative Dermatology, American Academy of Dermatology Association. The burden of skin diseases, 2005. Available from: http://www.lewin.com/~/media/lewin/site_sections/publications/april2005s.... Accessed March 12, 2012.
-
- Cockerell CJ, Wharton JR. New histopathological classification of actinic keratosis (incipient intraepidermal squamous cell carcinoma) J Drugs Dermatol. 2005;4(4):462–467. - PubMed
-
- Berman B, Bienstock L, Kuritzky L, Mayeaux EJ, Jr, Tyring SK. Actinic keratoses: sequelae and treatments. Recommendations from a consensus panel. J Fam Pract. 2006 May;55(Suppl 5):1–8. - PubMed
-
- Schwartz RA. The actinic keratosis. A perspective and update. Dermatol Surg. 1997;23(11):1009–1019. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

