Signal-mediated, AP-1/clathrin-dependent sorting of transmembrane receptors to the somatodendritic domain of hippocampal neurons
- PMID: 22958822
- PMCID: PMC3439821
- DOI: 10.1016/j.neuron.2012.07.007
Signal-mediated, AP-1/clathrin-dependent sorting of transmembrane receptors to the somatodendritic domain of hippocampal neurons
Abstract
Plasma membranes of the somatodendritic and axonal domains of neurons are known to have different protein compositions, but the molecular mechanisms that determine this polarized protein distribution remain poorly understood. Herein we show that somatodendritic sorting of various transmembrane receptors in rat hippocampal neurons is mediated by recognition of signals within the cytosolic domains of the proteins by the μ1A subunit of the adaptor protein-1 (AP-1) complex. This complex, in conjunction with clathrin, functions in the neuronal soma to exclude somatodendritic proteins from axonal transport carriers. Perturbation of this process affects dendritic spine morphology and decreases the number of synapses. These findings highlight the primary recognition event that underlies somatodendritic sorting and contribute to the evolving view of AP-1 as a global regulator of cell polarity.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
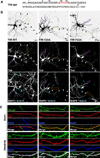

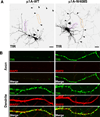

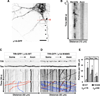
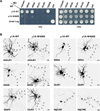


Comment in
-
The clathrin adaptor complex responsible for somatodendritic protein sorting.Neuron. 2012 Sep 6;75(5):742-4. doi: 10.1016/j.neuron.2012.08.023. Neuron. 2012. PMID: 22958815 Free PMC article.
References
-
- Arimura N, Kaibuchi K. Key regulators in neuronal polarity. Neuron. 2005;48:881–884. - PubMed
-
- Bae YK, Qin H, Knobel KM, Hu J, Rosenbaum JL, Barr MM. General and cell-type specific mechanisms target TRPP2/PKD-2 to cilia. Development. 2006;133:3859–3870. - PubMed
-
- Benhra N, Lallet S, Cotton M, Le Bras S, Dussert A, Le Borgne R. AP-1 controls the trafficking of Notch and Sanpodo toward E-cadherin junctions in sensory organ precursors. Curr Biol. 2011;21:87–95. - PubMed
-
- Bonifacino JS, Traub LM. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu Rev Biochem. 2003;72:395–447. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

