Temporal changes in PTEN and mTORC2 regulation of hematopoietic stem cell self-renewal and leukemia suppression
- PMID: 22958933
- PMCID: PMC3447536
- DOI: 10.1016/j.stem.2012.05.026
Temporal changes in PTEN and mTORC2 regulation of hematopoietic stem cell self-renewal and leukemia suppression
Abstract
Pten deletion from adult mouse hematopoietic cells activates the PI3-kinase pathway, inducing hematopoietic stem cell (HSC) proliferation, HSC depletion, and leukemogenesis. Pten is also mutated in human leukemias, but rarely in early childhood leukemias. We hypothesized that this reflects developmental changes in PI3-kinase pathway regulation. Here we show that Rictor deletion prevents leukemogenesis and HSC depletion after Pten deletion in adult mice, implicating mTORC2 activation in these processes. However, Rictor deletion had little effect on the function of normal HSCs. Moreover, Pten deletion from neonatal HSCs did not activate the PI3-kinase pathway or promote HSC proliferation, HSC depletion, or leukemogenesis. Pten is therefore required in adult, but not neonatal, HSCs to negatively regulate mTORC2 signaling. This demonstrates that some critical tumor suppressor mechanisms in adult cells are not required by neonatal cells. Developmental changes in key signaling pathways therefore confer temporal changes upon stem cell self-renewal and tumor suppressor mechanisms.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
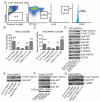
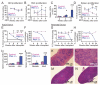
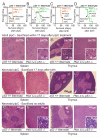
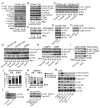


Comment in
-
Distinct and dynamic requirements for mTOR signaling in hematopoiesis and leukemogenesis.Cell Stem Cell. 2012 Sep 7;11(3):281-2. doi: 10.1016/j.stem.2012.08.007. Cell Stem Cell. 2012. PMID: 22958924
References
-
- Aiba Y, Yamazaki T, Okada T, Gotoh K, Sanjo H, Ogata M, Kurosaki T. BANK negatively regulates Akt activation and subsequent B cell responses. Immunity. 2006;24:259–268. - PubMed
-
- Clappier E, Cuccuini W, Kalota A, Crinquette A, Cayuela JM, Dik WA, Langerak AW, Montpellier B, Nadel B, Walrafen P, et al. The C-MYB locus is involved in chromosomal translocation and genomic duplications in human T-cell acute leukemia (T-ALL), the translocation defining a new T-ALL subtype in very young children. Blood. 2007;110:1251–1261. - PubMed
-
- Dahia PL, Aguiar RC, Alberta J, Kum JB, Caron S, Sill H, Marsh DJ, Ritz J, Freedman A, Stiles C, et al. PTEN is inversely correlated with the cell survival factor Akt/PKB and is inactivated via multiple mechanismsin haematological malignancies. Human Molecular Genetics. 1999;8:185–193. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

