Rediscovering Hydrogel-Based Double-Diffusion Systems for Studying Biomineralization
- PMID: 22962542
- PMCID: PMC3433082
- DOI: 10.1039/C2CE25289A
Rediscovering Hydrogel-Based Double-Diffusion Systems for Studying Biomineralization
Abstract
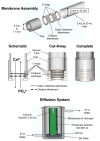
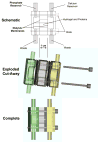
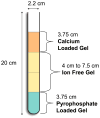
For those seeking to model biomineralization in vitro, hydrogels can serve as excellent models of the extracellular matrix (ECM) microenvironment. A major challenge posed in implementing such systems is the logistics involved, from fundamental engineering to experimental design. For the study of calcium phosphate (e.g., hydroxyapatite) formation, many researchers use hydrogel-based double-diffusion systems (DDSs). The various designs of these DDSs are seemingly as unique as their applications. In this Highlight, we present a survey of four distinct types of double-diffusion systems and evaluate them in the context of fundamental diffusion theory. Based upon this analysis, we present the design and evaluation of an optimized system. The techniques and framework for the evaluation and construction of a DDS presented here can be applied to any DDS that a researcher may want to implement for their particular studies of biomineralization.
Figures
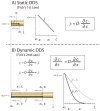




 )and calculated (
)and calculated (
 ) values of calcium and phosphate concentration in the end sections of two different lengths of diffusion tubes (6 cm and 8 cm) compared to the calculated value of c at x = L (
) values of calcium and phosphate concentration in the end sections of two different lengths of diffusion tubes (6 cm and 8 cm) compared to the calculated value of c at x = L (
 ) and the targeted theoretical value of 0.1% of c0 shown as lines. (
) and the targeted theoretical value of 0.1% of c0 shown as lines. (
 calcium,
calcium,
 phosphate). When the measured and calculated values are within error of each other, then the data are self-consistent and the assumed condition is considered valid (8 cm calcium and phosphate). When the measured value is equal to or less than the targeted value line, but the calculated value for c at x = L is greater than the targeted value line, the condition is considered quasi-infinite (not seen here). When both the calculated value 47 for c at x = L and the measured value at the end section are equal to or less than the targeted value line, then the condition can be considered semi-infinite (ex., calcium and phosphate 8 cm).
phosphate). When the measured and calculated values are within error of each other, then the data are self-consistent and the assumed condition is considered valid (8 cm calcium and phosphate). When the measured value is equal to or less than the targeted value line, but the calculated value for c at x = L is greater than the targeted value line, the condition is considered quasi-infinite (not seen here). When both the calculated value 47 for c at x = L and the measured value at the end section are equal to or less than the targeted value line, then the condition can be considered semi-infinite (ex., calcium and phosphate 8 cm).

 ) and phosphate (
) and phosphate (
 ) values for the optimized (condition D) and un-optimized (condition A) systems in the circulating semi-infinite reservoir design after 69.36 hours (2 ± 0.1 hours before precipitation was expected). Values for calcium and phosphate from the SD tubes in both conditions were statistically similar. The main difference between the two conditions is the measured calcium concentrations for the DD tubes in each case. (: The values for phosphate are within error of each other.)
) values for the optimized (condition D) and un-optimized (condition A) systems in the circulating semi-infinite reservoir design after 69.36 hours (2 ± 0.1 hours before precipitation was expected). Values for calcium and phosphate from the SD tubes in both conditions were statistically similar. The main difference between the two conditions is the measured calcium concentrations for the DD tubes in each case. (: The values for phosphate are within error of each other.)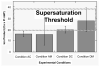
Similar articles
-
Bone ECM-inspired biomineralization chitin whisker liquid crystal hydrogels for bone regeneration.Int J Biol Macromol. 2023 Mar 15;231:123335. doi: 10.1016/j.ijbiomac.2023.123335. Epub 2023 Jan 20. Int J Biol Macromol. 2023. PMID: 36690237
-
Diffusion-Controllable Biomineralization Conducted In Situ in Hydrogels Based on Reversibly Cross-Linked Hyperbranched Polyglycidol.Biomacromolecules. 2017 Oct 9;18(10):3418-3431. doi: 10.1021/acs.biomac.7b01071. Epub 2017 Sep 13. Biomacromolecules. 2017. PMID: 28872843
-
A correlative spatiotemporal microscale study of calcium phosphate formation and transformation within an alginate hydrogel matrix.Acta Biomater. 2016 Oct 15;44:254-66. doi: 10.1016/j.actbio.2016.08.041. Epub 2016 Aug 24. Acta Biomater. 2016. PMID: 27567962
-
Enzymatic Approach in Calcium Phosphate Biomineralization: A Contribution to Reconcile the Physicochemical with the Physiological View.Int J Mol Sci. 2021 Nov 30;22(23):12957. doi: 10.3390/ijms222312957. Int J Mol Sci. 2021. PMID: 34884758 Free PMC article. Review.
-
Supramolecular Hydrogels Based on DNA Self-Assembly.Acc Chem Res. 2017 Apr 18;50(4):659-668. doi: 10.1021/acs.accounts.6b00524. Epub 2017 Mar 16. Acc Chem Res. 2017. PMID: 28299927 Review.
Cited by
-
Co-Deposition of a Hydrogel/Calcium Phosphate Hybrid Layer on 3D Printed Poly(Lactic Acid) Scaffolds via Dip Coating: Towards Automated Biomaterials Fabrication.Polymers (Basel). 2018 Mar 7;10(3):275. doi: 10.3390/polym10030275. Polymers (Basel). 2018. PMID: 30966310 Free PMC article.
-
Phosphorylation regulates the secondary structure and function of dentin phosphoprotein peptides.Bone. 2017 Feb;95:65-75. doi: 10.1016/j.bone.2016.10.028. Epub 2016 Nov 1. Bone. 2017. PMID: 27810285 Free PMC article.
-
Analytic Models of Oxygen and Nutrient Diffusion, Metabolism Dynamics, and Architecture Optimization in Three-Dimensional Tissue Constructs with Applications and Insights in Cerebral Organoids.Tissue Eng Part C Methods. 2016 Mar;22(3):221-49. doi: 10.1089/ten.TEC.2015.0375. Epub 2016 Jan 21. Tissue Eng Part C Methods. 2016. PMID: 26650970 Free PMC article.
-
Effect of the Electric Field on the Biomineralization of Collagen.Polymers (Basel). 2023 Jul 22;15(14):3121. doi: 10.3390/polym15143121. Polymers (Basel). 2023. PMID: 37514510 Free PMC article.
-
A Chitosan-Agarose Polysaccharide-Based Hydrogel for Biomimetic Remineralization of Dental Enamel.Biomolecules. 2021 Aug 2;11(8):1137. doi: 10.3390/biom11081137. Biomolecules. 2021. PMID: 34439803 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
