Advances in functional X-ray imaging techniques and contrast agents
- PMID: 22962667
- PMCID: PMC3569739
- DOI: 10.1039/c2cp41858d
Advances in functional X-ray imaging techniques and contrast agents
Abstract
X-rays have been used for non-invasive high-resolution imaging of thick biological specimens since their discovery in 1895. They are widely used for structural imaging of bone, metal implants, and cavities in soft tissue. Recently, a number of new contrast methodologies have emerged which are expanding X-ray's biomedical applications to functional as well as structural imaging. These techniques are promising to dramatically improve our ability to study in situ biochemistry and disease pathology. In this review, we discuss how X-ray absorption, X-ray fluorescence, and X-ray excited optical luminescence can be used for physiological, elemental, and molecular imaging of vasculature, tumors, pharmaceutical distribution, and the surface of implants. Imaging of endogenous elements, exogenous labels, and analytes detected with optical indicators will be discussed.
Figures






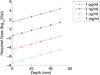


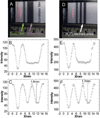

Similar articles
-
Surface modification effect on contrast agent efficiency for X-ray based spectral photon-counting scanner/luminescence imaging: from fundamental study to in vivo proof of concept.Nanoscale. 2024 Feb 8;16(6):2931-2944. doi: 10.1039/d3nr03710j. Nanoscale. 2024. PMID: 38230699
-
Review of in vivo optical molecular imaging and sensing from x-ray excitation.J Biomed Opt. 2021 Jan;26(1):010902. doi: 10.1117/1.JBO.26.1.010902. J Biomed Opt. 2021. PMID: 33386709 Free PMC article. Review.
-
[The Development of Luminescent Nano-probes on Hard X-ray Irradiation].Yakugaku Zasshi. 2016;136(1):17-20. doi: 10.1248/yakushi.15-00225-3. Yakugaku Zasshi. 2016. PMID: 26725662 Review. Japanese.
-
X-ray-induced shortwave infrared biomedical imaging using rare-earth nanoprobes.Nano Lett. 2015 Jan 14;15(1):96-102. doi: 10.1021/nl504123r. Epub 2014 Dec 15. Nano Lett. 2015. PMID: 25485705 Free PMC article.
-
Focused x-ray luminescence imaging system for small animals based on a rotary gantry.J Biomed Opt. 2021 Mar;26(3):036004. doi: 10.1117/1.JBO.26.3.036004. J Biomed Opt. 2021. PMID: 33738992 Free PMC article.
Cited by
-
Annealing-modulated nanoscintillators for nonconventional X-ray activation of comprehensive photodynamic effects in deep cancer theranostics.Theranostics. 2020 May 20;10(15):6758-6773. doi: 10.7150/thno.41752. eCollection 2020. Theranostics. 2020. PMID: 32550902 Free PMC article.
-
Imaging of Transmetallation and Chelation Phenomena Involving Radiological Contrast Agents in Mineral-Rich Fruits.Tomography. 2022 May 23;8(3):1413-1428. doi: 10.3390/tomography8030114. Tomography. 2022. PMID: 35645400 Free PMC article.
-
Hard X-ray-induced optical luminescence via biomolecule-directed metal clusters.Chem Commun (Camb). 2014 Apr 7;50(27):3549-51. doi: 10.1039/c3cc48661c. Chem Commun (Camb). 2014. PMID: 24463467 Free PMC article.
-
Nanoscintillator-mediated X-ray inducible photodynamic therapy for in vivo cancer treatment.Nano Lett. 2015 Apr 8;15(4):2249-56. doi: 10.1021/nl504044p. Epub 2015 Mar 12. Nano Lett. 2015. PMID: 25756781 Free PMC article.
-
Chemotherapy and Physical Therapeutics Modulate Antigens on Cancer Cells.Front Immunol. 2022 Jul 6;13:889950. doi: 10.3389/fimmu.2022.889950. eCollection 2022. Front Immunol. 2022. PMID: 35874714 Free PMC article. Review.
References
-
- Ono M. Chemical and Pharmaceutical Bulletin. 2009;57:1029–1039. - PubMed
-
- Rahmim A, Zaidi H. Nuclear medicine communications. 2008;29:193. - PubMed
-
- Levin CS, Hoffman EJ. Phys. Med. Bio. 1999;44:781. - PubMed
-
- Derenzo SE, Moses WW, Huesman RH, Budinger TF. Annals of nuclear medicine. 1993;7:3–3.
-
- Kojima A, Matsumoto M, Takahashi M, Hirota Y, Yoshida H. Journal of nuclear medicine: official publication, Society of Nuclear Medicine. 1989;30:508. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

