HLA-DM constrains epitope selection in the human CD4 T cell response to vaccinia virus by favoring the presentation of peptides with longer HLA-DM-mediated half-lives
- PMID: 22966084
- PMCID: PMC3466378
- DOI: 10.4049/jimmunol.1200626
HLA-DM constrains epitope selection in the human CD4 T cell response to vaccinia virus by favoring the presentation of peptides with longer HLA-DM-mediated half-lives
Abstract
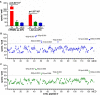
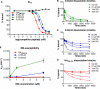
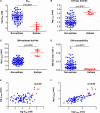
HLA-DM (DM) is a nonclassical MHC class II (MHC II) protein that acts as a peptide editor to mediate the exchange of peptides loaded onto MHC II during Ag presentation. Although the ability of DM to promote peptide exchange in vitro and in vivo is well established, the role of DM in epitope selection is still unclear, especially in human response to infectious disease. In this study, we addressed this question in the context of the human CD4 T cell response to vaccinia virus. We measured the IC(50), intrinsic dissociation t(1/2), and DM-mediated dissociation t(1/2) for a large set of peptides derived from the major core protein A10L and other known vaccinia epitopes bound to HLA-DR1 and compared these properties to the presence and magnitude of peptide-specific CD4(+) T cell responses. We found that MHC II-peptide complex kinetic stability in the presence of DM distinguishes T cell epitopes from nonrecognized peptides in A10L peptides and also in a set of predicted tight binders from the entire vaccinia genome. Taken together, these analyses demonstrate that DM-mediated dissociation t(1/2) is a strong and independent factor governing peptide immunogenicity by favoring the presentation of peptides with greater kinetic stability in the presence of DM.
Figures



Similar articles
-
A novel method to measure HLA-DM-susceptibility of peptides bound to MHC class II molecules based on peptide binding competition assay and differential IC(50) determination.J Immunol Methods. 2014 Apr;406:21-33. doi: 10.1016/j.jim.2014.02.008. Epub 2014 Feb 25. J Immunol Methods. 2014. PMID: 24583195 Free PMC article.
-
Human CD4+ T cell epitopes from vaccinia virus induced by vaccination or infection.PLoS Pathog. 2007 Oct 12;3(10):1511-29. doi: 10.1371/journal.ppat.0030144. PLoS Pathog. 2007. PMID: 17937498 Free PMC article.
-
Vaccinia peptides eluted from HLA-DR1 isolated from virus-infected cells are recognized by CD4+ T cells from a vaccinated donor.J Proteome Res. 2008 Jul;7(7):2703-11. doi: 10.1021/pr700780x. Epub 2008 May 29. J Proteome Res. 2008. PMID: 18507432 Free PMC article.
-
Mechanisms of peptide repertoire selection by HLA-DM.Trends Immunol. 2013 Oct;34(10):495-501. doi: 10.1016/j.it.2013.06.002. Epub 2013 Jul 5. Trends Immunol. 2013. PMID: 23835076 Free PMC article. Review.
-
Revisiting nonclassical HLA II functions in antigen presentation: Peptide editing and its modulation.HLA. 2020 Oct;96(4):415-429. doi: 10.1111/tan.14007. Epub 2020 Aug 27. HLA. 2020. PMID: 32767512 Review.
Cited by
-
An unstable Th epitope of P. falciparum fosters central memory T cells and anti-CS antibody responses.PLoS One. 2014 Jul 1;9(7):e100639. doi: 10.1371/journal.pone.0100639. eCollection 2014. PLoS One. 2014. PMID: 24983460 Free PMC article.
-
The Dendritic Cell Major Histocompatibility Complex II (MHC II) Peptidome Derives from a Variety of Processing Pathways and Includes Peptides with a Broad Spectrum of HLA-DM Sensitivity.J Biol Chem. 2016 Mar 11;291(11):5576-5595. doi: 10.1074/jbc.M115.655738. Epub 2016 Jan 6. J Biol Chem. 2016. PMID: 26740625 Free PMC article.
-
Distorted Immunodominance by Linker Sequences or other Epitopes from a Second Protein Antigen During Antigen-Processing.Sci Rep. 2017 Apr 19;7:46418. doi: 10.1038/srep46418. Sci Rep. 2017. PMID: 28422163 Free PMC article.
-
HLA-DM and HLA-DO, key regulators of MHC-II processing and presentation.Curr Opin Immunol. 2014 Feb;26:115-22. doi: 10.1016/j.coi.2013.11.005. Epub 2013 Dec 8. Curr Opin Immunol. 2014. PMID: 24463216 Free PMC article. Review.
-
Protein-Specific Features Associated with Variability in Human Antibody Responses to Plasmodium falciparum Malaria Antigens.Am J Trop Med Hyg. 2018 Jan;98(1):57-66. doi: 10.4269/ajtmh.17-0437. Am J Trop Med Hyg. 2018. PMID: 29141757 Free PMC article.
References
-
- Germain RN. MHC-dependent antigen processing and peptide presentation: providing ligands for T lymphocyte activation. Cell. 1994;76:287–299. - PubMed
-
- Bakke O, Dobberstein B. MHC class II-associated invariant chain contains a sorting signal for endosomal compartments. Cell. 1990;63:707–716. - PubMed
-
- Roche PA, Cresswell P. Invariant chain association with HLA-DR molecules inhibits immunogenic peptide binding. Nature. 1990;345:615–618. - PubMed
-
- Sloan VS, Cameron P, Porter G, Gammon M, Amaya M, Mellins E, Zaller DM. Mediation by HLA-DM of dissociation of peptides from HLA-DR. Nature. 1995;375:802–806. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

