Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura
- PMID: 22971321
- PMCID: PMC3489865
- DOI: 10.1186/1744-8069-8-66
Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura
Abstract
Background: Migraine and other headache disorders affect a large percentage of the population and cause debilitating pain. Activation and sensitization of the trigeminal primary afferent neurons innervating the dura and cerebral vessels is a crucial step in the "headache circuit". Many dural afferent neurons respond to algesic and inflammatory agents. Given the clear role of the transient receptor potential (TRP) family of channels in both sensing chemical stimulants and mediating inflammatory pain, we investigated the expression of TRP channels in dural afferent neurons.
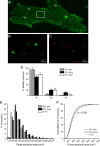
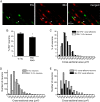
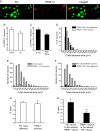
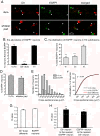
Methods: We used two fluorescent tracers to retrogradely label dural afferent neurons in adult mice and quantified the abundance of peptidergic and non-peptidergic neuron populations using calcitonin gene-related peptide immunoreactivity (CGRP-ir) and isolectin B4 (IB4) binding as markers, respectively. Using immunohistochemistry, we compared the expression of TRPV1 and TRPA1 channels in dural afferent neurons with the expression in total trigeminal ganglion (TG) neurons. To examine the distribution of TRPM8 channels, we labeled dural afferent neurons in mice expressing farnesylated enhanced green fluorescent protein (EGFPf) from a TRPM8 locus. We used nearest-neighbor measurement to predict the spatial association between dural afferent neurons and neurons expressing TRPA1 or TRPM8 channels in the TG.
Results and conclusions: We report that the size of dural afferent neurons is significantly larger than that of total TG neurons and facial skin afferents. Approximately 40% of dural afferent neurons exhibit IB4 binding. Surprisingly, the percentage of dural afferent neurons containing CGRP-ir is significantly lower than those of total TG neurons and facial skin afferents. Both TRPV1 and TRPA1 channels are expressed in dural afferent neurons. Furthermore, nearest-neighbor measurement indicates that TRPA1-expressing neurons are clustered around a subset of dural afferent neurons. Interestingly, TRPM8-expressing neurons are virtually absent in the dural afferent population, nor do these neurons cluster around dural afferent neurons. Taken together, our results suggest that TRPV1 and TRPA1 but not TRPM8 channels likely contribute to the excitation of dural afferent neurons and the subsequent activation of the headache circuit. These results provide an anatomical basis for understanding further the functional significance of TRP channels in headache pathophysiology.
Figures



Similar articles
-
Function and postnatal changes of dural afferent fibers expressing TRPM8 channels.Mol Pain. 2015 Jun 26;11:37. doi: 10.1186/s12990-015-0043-0. Mol Pain. 2015. PMID: 26111800 Free PMC article.
-
Quantitative Analysis of Mouse Dural Afferent Neurons Expressing TRPM8, VGLUT3, and NF200.Headache. 2018 Jan;58(1):88-101. doi: 10.1111/head.13188. Epub 2017 Sep 19. Headache. 2018. PMID: 28925503 Free PMC article.
-
Distinct expression of TRPM8, TRPA1, and TRPV1 mRNAs in rat primary afferent neurons with adelta/c-fibers and colocalization with trk receptors.J Comp Neurol. 2005 Dec 26;493(4):596-606. doi: 10.1002/cne.20794. J Comp Neurol. 2005. PMID: 16304633
-
Targeting TRP channels for novel migraine therapeutics.ACS Chem Neurosci. 2014 Nov 19;5(11):1085-96. doi: 10.1021/cn500083e. Epub 2014 Aug 29. ACS Chem Neurosci. 2014. PMID: 25138211 Free PMC article. Review.
-
The role of chemosensitive afferent nerves and TRP ion channels in the pathomechanism of headaches.Pflugers Arch. 2012 Sep;464(3):239-48. doi: 10.1007/s00424-012-1142-7. Epub 2012 Aug 9. Pflugers Arch. 2012. PMID: 22875278 Review.
Cited by
-
Function and postnatal changes of dural afferent fibers expressing TRPM8 channels.Mol Pain. 2015 Jun 26;11:37. doi: 10.1186/s12990-015-0043-0. Mol Pain. 2015. PMID: 26111800 Free PMC article.
-
TRPM8 and Migraine.Headache. 2016 Oct;56(9):1406-1417. doi: 10.1111/head.12948. Epub 2016 Sep 16. Headache. 2016. PMID: 27634619 Free PMC article. Review.
-
Functional analysis of a migraine-associated TRESK K+ channel mutation.J Neurosci. 2013 Jul 31;33(31):12810-24. doi: 10.1523/JNEUROSCI.1237-13.2013. J Neurosci. 2013. PMID: 23904616 Free PMC article.
-
Over-expression of TRESK K(+) channels reduces the excitability of trigeminal ganglion nociceptors.PLoS One. 2014 Jan 23;9(1):e87029. doi: 10.1371/journal.pone.0087029. eCollection 2014. PLoS One. 2014. PMID: 24466320 Free PMC article.
-
Ethyl Vanillin Activates TRPA1.J Pharmacol Exp Ther. 2017 Sep;362(3):368-377. doi: 10.1124/jpet.116.239384. Epub 2017 Jun 15. J Pharmacol Exp Ther. 2017. PMID: 28620120 Free PMC article.
References
-
- Moskowitz MA. Pathophysiology of headache–past and present. Headache. 2007;47(Suppl 1):S58–S63. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

