Cloning and expression of porcine Colony Stimulating Factor-1 (CSF-1) and Colony Stimulating Factor-1 Receptor (CSF-1R) and analysis of the species specificity of stimulation by CSF-1 and Interleukin 34
- PMID: 22974529
- PMCID: PMC3500696
- DOI: 10.1016/j.cyto.2012.08.008
Cloning and expression of porcine Colony Stimulating Factor-1 (CSF-1) and Colony Stimulating Factor-1 Receptor (CSF-1R) and analysis of the species specificity of stimulation by CSF-1 and Interleukin 34
Abstract
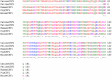
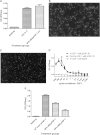
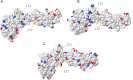
Macrophage Colony Stimulating Factor (CSF-1) controls the survival, differentiation and proliferation of cells of the mononuclear phagocyte system. A second ligand for the CSF-1R, Interleukin 34 (IL-34), has been described, but its physiological role is not yet known. The domestic pig provides an alternative to traditional rodent models for evaluating potential therapeutic applications of CSF-1R agonists and antagonists. To enable such studies, we cloned and expressed active pig CSF-1. To provide a bioassay, pig CSF-1R was expressed in the factor-dependent Ba/F3 cell line. On this transfected cell line, recombinant porcine CSF-1 and human CSF-1 had identical activity. Mouse CSF-1 does not interact with the human CSF-1 receptor but was active on pig. By contrast, porcine CSF-1 was active on mouse, human, cat and dog cells. IL-34 was previously shown to be species-specific, with mouse and human proteins demonstrating limited cross-species activity. The pig CSF-1R was equally responsive to both mouse and human IL-34. Based upon the published crystal structures of CSF-1/CSF-1R and IL34/CSF-1R complexes, we discuss the molecular basis for the species specificity.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures






References
-
- Chitu V., Stanley E.R. Colony-stimulating factor-1 in immunity and inflammation. Curr Opin Immunol. 2006;18:39–48. - PubMed
-
- Sweet M.J., Hume D.A. CSF-1 as a regulator of macrophage activation and immune responses. Arch Immunol Ther Exp. 2003;51:169–177. - PubMed
-
- Sherr C.J., Rettenmier C.W. The fms gene and the CSF-1 receptor. Cancer Surv. 1986;5:221–232. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- BB/I013113/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BBS/E/D/20251969/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- G0901193/MRC_/Medical Research Council/United Kingdom
- 338BCB R40954/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

