Non-physiological amino acid (NPAA) therapy targeting brain phenylalanine reduction: pilot studies in PAHENU2 mice
- PMID: 22976763
- PMCID: PMC3654543
- DOI: 10.1007/s10545-012-9524-8
Non-physiological amino acid (NPAA) therapy targeting brain phenylalanine reduction: pilot studies in PAHENU2 mice
Abstract
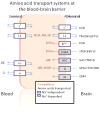
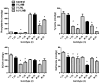
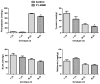
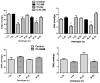
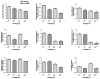
Transport of large neutral amino acids (LNAA) across the blood brain barrier (BBB) is facilitated by the L-type amino acid transporter, LAT1. Peripheral accumulation of one LNAA (e.g., phenylalanine (phe) in PKU) is predicted to increase uptake of the offending amino acid to the detriment of others, resulting in disruption of brain amino acid homeostasis. We hypothesized that selected non-physiological amino acids (NPAAs) such as DL-norleucine (NL), 2-aminonorbornane (NB; 2-aminobicyclo-(2,1,1)-heptane-2-carboxylic acid), 2-aminoisobutyrate (AIB), and N-methyl-aminoisobutyrate (MAIB), acting as competitive inhibitors of various brain amino acid transporters, could reduce brain phe in Pah (enu2) mice, a relevant murine model of PKU. Oral feeding of 5 % NL, 5 % AIB, 0.5 % NB and 3 % MAIB reduced brain phe by 56 % (p < 0.01), -1 % (p = NS), 27 % (p < 0.05) and 14 % (p < 0.01), respectively, compared to untreated subjects. Significant effects on other LNAAs (tyrosine, methionine, branched chain amino acids) were also observed, however, with MAIB displaying the mildest effects. Of interest, MAIB represents an inhibitor of the system A (alanine) transporter that primarily traffics small amino acids and not LNAAs. Our studies represent the first in vivo use of these NPAAs in Pah (enu2) mice, and provide proof-of-principle for their further preclinical development, with the long-term objective of identifying NPAA combinations and concentrations that selectively restrict brain phe transport while minimally impacting other LNAAs and downstream intermediates.
Figures
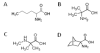
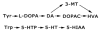
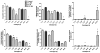
References
-
- Arning E, Bottiglieri T, Sun Q, et al. Metabolic profiling in phenylalanine hydroxylase-deficient (Pah−/−) mouse brain reveals decreased amino acid neurotransmitters and preferential alterations of the serotoninergic system. Molec Genet Metab. 2009;98:21.
-
- Babu E, Kanai Y, Chairoungdua A, et al. Identification of a novel system L amino acid transporter structurally distinct from heterodimeric amino acid transporters. J Biol Chem. 2003;278:43838–45. - PubMed
-
- Bodoy S, Martín L, Zorzano A, Palacín M, Estévez R, Bertran J. Identification of LAT4, a novel amino acid transporter with system L activity. J Biol Chem. 2005;280:12002–11. - PubMed
-
- Bröer S, Brookes N. Transfer of glutamine between astrocytes and neurons. J Neurochem. 2001;77:705–19. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

