Intravesical administration of pirarubicin against superficial bladder cancer: Relationship between tumor tissue concentration and exposure time in the bladder or therapeutic effect
- PMID: 22977595
- PMCID: PMC3440819
- DOI: 10.3892/etm.2011.315
Intravesical administration of pirarubicin against superficial bladder cancer: Relationship between tumor tissue concentration and exposure time in the bladder or therapeutic effect
Abstract
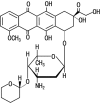
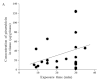
The aim of this study was to investigate the relationship between tissue concentrations and exposure times or therapeutic effect of an anthracycline anticancer drug, pirarubicin, in bladder cancer tissue after single intravesical administration against superficial bladder cancer. The concentrations of pirarubicin in tumor tissues and serum were measured at designated collection times after a single intravesical administration of pirarubicin (30 mg) in 22 patients with superficial bladder cancer. A wide range of concentrations of pirarubicin in bladder cancer tissue was observed (2.3-125 μg/g of tissue), although serum pirarubicin concentrations were not detected in any of the patients. Recurrence of superficial bladder cancer after transurethral resection of the bladder tumor (TUR-BT) was observed in 2 patients (9%). The concentration of pirarubicin in the tumor tissue tended to be higher as the exposure time increased. There was a weak relationship between the pirarubicin tissue concentration and tumor size. However, no significant relationship between tissue pirarubicin concentrations and the prophylactic effect against intravesical recurrence of bladder cancer after TUR-BT was observed. All patients had no adverse events, such as bladder irritation and local toxicity, caused by the treatment with pirarubicin. These findings suggest that prior to single intravesical administration of pirarubicin to patients with superficial bladder cancer the exposure time and tumor size should be considered.
Figures

References
-
- Lutzeyer W, Rubben H, Dahm H. Prognostic parameters in superficial bladder cancer: an analysis of 315 cases. J Urol. 1982;127:250–252. - PubMed
-
- Herr HW, Laudone VP, Whitmore WF. An overview of intravesical therapy for superficial bladder tumors. J Urol. 1987;138:1363–1368. - PubMed
-
- Miki T, Nonomura N, Kojima Y, et al. A randomized study on intravesical pirarubicin (THP) chemoprophylaxis of recurrence after transurethral resection of superficial bladder cancer. Hinyokika Kiyo. 1997;43:907–912. (In Japanese). - PubMed
-
- Sylvester RJ, van der MA, Lamm DL. Intravesical bacillus Calmette-Guerin reduces the risk of progression in patients with superficial bladder cancer: a meta-analysis of the published results of randomized clinical trials. J Urol. 2002;168:1964–1970. - PubMed
-
- Lamm DL, Blumenstein BA, Crawford ED, et al. A randomized trial of intravesical doxorubicin and immunotherapy with bacille Calmette-Guérin for transitional-cell carcinoma of the bladder. N Engl J Med. 1991;325:1205–1209. - PubMed
LinkOut - more resources
Full Text Sources
