Flexible trial design in practice - stopping arms for lack-of-benefit and adding research arms mid-trial in STAMPEDE: a multi-arm multi-stage randomized controlled trial
- PMID: 22978443
- PMCID: PMC3466132
- DOI: 10.1186/1745-6215-13-168
Flexible trial design in practice - stopping arms for lack-of-benefit and adding research arms mid-trial in STAMPEDE: a multi-arm multi-stage randomized controlled trial
Abstract
Background: Systemic Therapy for Advanced or Metastatic Prostate cancer: Evaluation of Drug Efficacy (STAMPEDE) is a randomized controlled trial that follows a novel multi-arm, multi-stage (MAMS) design. We describe methodological and practical issues arising with (1) stopping recruitment to research arms following a pre-planned intermediate analysis and (2) adding a new research arm during the trial.
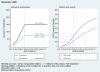
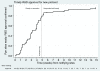
Methods: STAMPEDE recruits men who have locally advanced or metastatic prostate cancer who are starting standard long-term hormone therapy. Originally there were five research and one control arms, each undergoing a pilot stage (focus: safety, feasibility), three intermediate 'activity' stages (focus: failure-free survival), and a final 'efficacy' stage (focus: overall survival). Lack-of-sufficient-activity guidelines support the pairwise interim comparisons of each research arm against the control arm; these pre-defined activity cut-off becomes increasingly stringent over the stages. Accrual of further patients continues to the control arm and to those research arms showing activity and an acceptable safety profile. The design facilitates adding new research arms should sufficiently interesting agents emerge. These new arms are compared only to contemporaneously recruited control arm patients using the same intermediate guidelines in a time-delayed manner. The addition of new research arms is subject to adequate recruitment rates to support the overall trial aims.
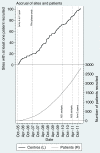
Results: (1) Stopping Existing Therapy: After the second intermediate activity analysis, recruitment was discontinued to two research arms for lack-of-sufficient activity. Detailed preparations meant that changes were implemented swiftly at 100 international centers and recruitment continued seamlessly into Activity Stage III with 3 remaining research arms and the control arm. Further regulatory and ethical approvals were not required because this was already included in the initial trial design.(2) Adding New Therapy: An application to add a new research arm was approved by the funder, (who also organized peer review), industrial partner and regulatory and ethical bodies. This was all done in advance of any decision to stop current therapies.
Conclusions: The STAMPEDE experience shows that recruitment to a MAMS trial and mid-flow changes its design are achievable with good planning. This benefits patients and the scientific community as research treatments are evaluated in a more efficient and cost-effective manner.
Trial registration: ISRCTN78818544, NCT00268476. First patient into trial: 17 October 2005. First patient into abiraterone comparison: 15 November 2011.
Figures


References
-
- James N, Sydes M, Clarke N, Mason M, Dearnaley D, Anderson J, Popert R, Sanders K, Morgan R, Stansfeld J, Dwyer J, Masters J, Parmar MKB. STAMPEDE: systemic therapy for advancing or metastatic prostate cancer - a multi-Arm multi-stage randomised controlled trial. Clin Onc. 2008;20:577–581. doi: 10.1016/j.clon.2008.07.002. - DOI - PubMed
-
- James ND, Sydes MR, Clarke NW, Mason MD, Dearnaley DP, Anderson J, Popert RJ, Sanders K, Morgan RC, Stansfeld J, Dwyer J, Masters J, Parmar MKB. Systemic therapy for advancing or metastatic prostate cancer (STAMPEDE): a multi-arm, multistage randomized controlled trial. BJU Int. 2009;103:464–469. doi: 10.1111/j.1464-410X.2008.08034.x. - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

