Inhibition of NaV1.6 sodium channel currents by a novel series of 1,4-disubstituted-triazole derivatives obtained via copper-catalyzed click chemistry
- PMID: 22981330
- PMCID: PMC3475177
- DOI: 10.1016/j.bmcl.2012.08.067
Inhibition of NaV1.6 sodium channel currents by a novel series of 1,4-disubstituted-triazole derivatives obtained via copper-catalyzed click chemistry
Abstract
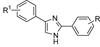
We have synthesized and evaluated a series of 1,4-disubstituted-triazole derivatives for inhibition of the rat Na(V)1.6 sodium channel isoform, an isoform thought to play an important role in controlling neuronal firing. Starting from a series of 2,4(1H)-diarylimidazoles previously published, we decided to extend the SAR study by replacing the imidazole with a different heterocyclic scaffold and by varying the aryl substituents on the central aromatic ring. The 1,4-disubstituted 1,2,3-triazoles were prepared employing the copper-catalyzed azide-alkyne cycloaddition (CuAAC). Many of the new molecules were able to block the rNa(v)1.6 currents at 10 μM by over 20%, displaying IC(50) values ranging in the low micromolar, thus indicating that triazole can efficiently replace the central heterocyclic core. Moreover, the introduction of a long chain at C4 of the central triazole seems beneficial for increased rNa(v)1.6 current block, whereas the length of N1 substituent seems less crucial for inhibition, as long as a phenyl ring is not direcly connected to the triazole. These results provide additional information on the structural features necessary for block of the voltage-gated sodium channels. These new data will be exploited in the preparation of new compounds and could result in potentially useful AEDs.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Click chemistry beyond metal-catalyzed cycloaddition as a remarkable tool for green chemical synthesis of antifungal medications.Chem Biol Drug Des. 2024 Jun;103(6):e14555. doi: 10.1111/cbdd.14555. Chem Biol Drug Des. 2024. PMID: 38862260 Review.
-
Synthesis of 6-amino-5-cyano-1,4-disubstituted-2(1H)-pyrimidinones via copper-(I)-catalyzed alkyne-azide 'click chemistry' and their reactivity.Molecules. 2010 Dec 3;15(12):8841-55. doi: 10.3390/molecules1512884. Molecules. 2010. PMID: 21131903 Free PMC article.
-
Design and synthesis of 2'-deoxy-2'-[(1,2,3)triazol-1-yl]uridines using click chemistry approach.Nucleosides Nucleotides Nucleic Acids. 2015;34(5):371-8. doi: 10.1080/15257770.2014.1003652. Nucleosides Nucleotides Nucleic Acids. 2015. PMID: 25874945
-
Synthesis of glycoconjugate mimics by 'click chemistry'.Carbohydr Res. 2019 Oct 1;484:107775. doi: 10.1016/j.carres.2019.107775. Epub 2019 Aug 9. Carbohydr Res. 2019. PMID: 31430604
-
An Overview on the Performance of 1,2,3-Triazole Derivatives as Corrosion Inhibitors for Metal Surfaces.Int J Mol Sci. 2021 Dec 21;23(1):16. doi: 10.3390/ijms23010016. Int J Mol Sci. 2021. PMID: 35008481 Free PMC article. Review.
Cited by
-
Recent developments on triazole nucleus in anticonvulsant compounds: a review.J Enzyme Inhib Med Chem. 2018 Dec;33(1):453-478. doi: 10.1080/14756366.2017.1423068. J Enzyme Inhib Med Chem. 2018. PMID: 29383949 Free PMC article. Review.
-
Cryo-EM structure of human voltage-gated sodium channel Nav1.6.Proc Natl Acad Sci U S A. 2023 Jan 31;120(5):e2220578120. doi: 10.1073/pnas.2220578120. Epub 2023 Jan 25. Proc Natl Acad Sci U S A. 2023. PMID: 36696443 Free PMC article.
-
Catalytic activity and mechanistic investigation of 1D 2-Picolinic acid based Cu(II) coordination polymer in the selective construction of 1,4-disubstituted triazoles.Sci Rep. 2022 Aug 26;12(1):14613. doi: 10.1038/s41598-022-18780-x. Sci Rep. 2022. PMID: 36028653 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Miscellaneous

