New insight into the ochratoxin A biosynthetic pathway through deletion of a nonribosomal peptide synthetase gene in Aspergillus carbonarius
- PMID: 22983973
- PMCID: PMC3497364
- DOI: 10.1128/AEM.02508-12
New insight into the ochratoxin A biosynthetic pathway through deletion of a nonribosomal peptide synthetase gene in Aspergillus carbonarius
Abstract
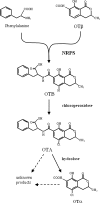
Ochratoxin A (OTA), a mycotoxin produced by Aspergillus and Penicillium species, is composed of a dihydroisocoumarin ring linked to phenylalanine, and its biosynthetic pathway has not yet been completely elucidated. Most of the knowledge regarding the genetic and enzymatic aspects of OTA biosynthesis has been elucidated in Penicillium species. In Aspergillus species, only pks genes involved in the initial steps of the pathway have been partially characterized. In our study, the inactivation of a gene encoding a nonribosomal peptide synthetase (NRPS) in OTA-producing A. carbonarius ITEM 5010 has eliminated the ability of this fungus to produce OTA. This is the first report on the involvement of an nrps gene product in OTA biosynthetic pathway in an Aspergillus species. The absence of OTA and ochratoxin α, the isocoumaric derivative of OTA, and the concomitant increase of ochratoxin β, the dechloro analog of ochratoxin α, were observed in the liquid culture of transformed strain. The data provide the first evidence that the enzymatic step adding phenylalanine to polyketide dihydroisocoumarin precedes the chlorination step to form OTA in A. carbonarius and that ochratoxin α is a product of hydrolysis of OTA, giving an interesting new insight into the biosynthetic pathway of the toxin.
Figures


Similar articles
-
Identification of a Halogenase Involved in the Biosynthesis of Ochratoxin A in Aspergillus carbonarius.Appl Environ Microbiol. 2016 Aug 30;82(18):5631-41. doi: 10.1128/AEM.01209-16. Print 2016 Sep 15. Appl Environ Microbiol. 2016. PMID: 27422838 Free PMC article.
-
A Consensus Ochratoxin A Biosynthetic Pathway: Insights from the Genome Sequence of Aspergillus ochraceus and a Comparative Genomic Analysis.Appl Environ Microbiol. 2018 Sep 17;84(19):e01009-18. doi: 10.1128/AEM.01009-18. Print 2018 Oct 1. Appl Environ Microbiol. 2018. PMID: 30054361 Free PMC article.
-
Identification and characterization of the polyketide synthase involved in ochratoxin A biosynthesis in Aspergillus carbonarius.Int J Food Microbiol. 2014 Jun 2;179:10-7. doi: 10.1016/j.ijfoodmicro.2014.03.013. Epub 2014 Mar 18. Int J Food Microbiol. 2014. PMID: 24699234
-
Ochratoxin A Producing Fungi, Biosynthetic Pathway and Regulatory Mechanisms.Toxins (Basel). 2016 Mar 21;8(3):83. doi: 10.3390/toxins8030083. Toxins (Basel). 2016. PMID: 27007394 Free PMC article. Review.
-
Advances in the molecular diagnosis of ochratoxin A-producing fungi.Food Addit Contam. 2005 Apr;22(4):324-34. doi: 10.1080/02652030500058452. Food Addit Contam. 2005. PMID: 16019802 Review.
Cited by
-
Phylogenetic study of polyketide synthases and nonribosomal peptide synthetases involved in the biosynthesis of mycotoxins.Toxins (Basel). 2013 Apr 19;5(4):717-42. doi: 10.3390/toxins5040717. Toxins (Basel). 2013. PMID: 23604065 Free PMC article.
-
Unlocking the Transcriptional Reprogramming Repertoire between Variety-Dependent Responses of Grapevine Berries to Infection by Aspergillus carbonarius.Plants (Basel). 2024 Jul 25;13(15):2043. doi: 10.3390/plants13152043. Plants (Basel). 2024. PMID: 39124161 Free PMC article.
-
Production of Bio-Based Pigments from Food Processing Industry By-Products (Apple, Pomegranate, Black Carrot, Red Beet Pulps) Using Aspergillus carbonarius.J Fungi (Basel). 2020 Oct 22;6(4):240. doi: 10.3390/jof6040240. J Fungi (Basel). 2020. PMID: 33105686 Free PMC article.
-
Beyond the Biosynthetic Gene Cluster Paradigm: Genome-Wide Coexpression Networks Connect Clustered and Unclustered Transcription Factors to Secondary Metabolic Pathways.Microbiol Spectr. 2021 Oct 31;9(2):e0089821. doi: 10.1128/Spectrum.00898-21. Epub 2021 Sep 15. Microbiol Spectr. 2021. PMID: 34523946 Free PMC article.
-
Benzyl isothiocyanate fumigation inhibits growth, membrane integrity and mycotoxin production in Alternaria alternata.RSC Adv. 2020 Jan 8;10(3):1829-1837. doi: 10.1039/c9ra09225k. eCollection 2020 Jan 7. RSC Adv. 2020. PMID: 35494694 Free PMC article.
References
-
- Abouzied MM, et al. 2002. Ochratoxin A concentrations in food and feed from a region with Balkan endemic nephropathy. Food Addit. Contam. 19:755–764 - PubMed
-
- Abrunhosa L, Serra R, Venancio A. 2002. Biodegradation of ochratoxin A by fungi isolate from grapes. J. Agric. Food Chem. 50:7493–7496 - PubMed
-
- Abrunhosa L, Venancio EA. 2007. Isolation and purification of an enzyme hydrolyzing ochratoxin A from Aspergillus niger. Biotechnol. Lett. 29:1909–1914 - PubMed
-
- Atoui A, Dao HP, Mathieu F, Lebrihi A. 2006. Amplification and diversity analysis of ketosynthase domains of putative polyketide synthase genes in Aspergillus ochraceus and Aspergillus carbonarius producers of ochratoxin A. Mol. Nutr. Food Res. 50:488–493 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

