Multi-scale biomechanical remodeling in aging and genetic mutant murine mitral valve leaflets: insights into Marfan syndrome
- PMID: 22984535
- PMCID: PMC3439411
- DOI: 10.1371/journal.pone.0044639
Multi-scale biomechanical remodeling in aging and genetic mutant murine mitral valve leaflets: insights into Marfan syndrome
Abstract
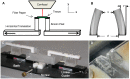
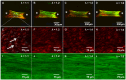
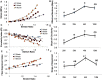
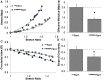
Mitral valve degeneration is a key component of the pathophysiology of Marfan syndrome. The biomechanical consequences of aging and genetic mutation in mitral valves are poorly understood because of limited tools to study this in mouse models. Our aim was to determine the global biomechanical and local cell-matrix deformation relationships in the aging and Marfan related Fbn1 mutated murine mitral valve. To conduct this investigation, a novel stretching apparatus and gripping method was implemented to directly quantify both global tissue biomechanics and local cellular deformation and matrix fiber realignment in murine mitral valves. Excised mitral valve leaflets from wild-type and Fbn1 mutant mice from 2 weeks to 10 months in age were tested in circumferential orientation under continuous laser optical imaging. Mouse mitral valves stiffen with age, correlating with increases in collagen fraction and matrix fiber alignment. Fbn1 mutation resulted in significantly more compliant valves (modulus 1.34 ± 0.12 vs. 2.51 ± 0.31 MPa, respectively, P<.01) at 4 months, corresponding with an increase in proportion of GAGs and decrease in elastin fraction. Local cellular deformation and fiber alignment change linearly with global tissue stretch, and these slopes become more extreme with aging. In comparison, Fbn1 mutated valves have decoupled cellular deformation and fiber alignment with tissue stretch. Taken together, quantitative understanding of multi-scale murine planar tissue biomechanics is essential for establishing consequences of aging and genetic mutations. Decoupling of local cell-matrix deformation kinematics with global tissue stretch may be an important mechanism of normal and pathological biomechanical remodeling in valves.
Conflict of interest statement
Figures


References
-
- Pyeritz RE (2000) The Marfan syndrome. Annu Rev Med 51: 481–510. - PubMed
-
- Dietz HC, Loeys B, Carta L, Ramirez F (2005) Recent progress towards a molecular understanding of Marfan syndrome. Am J Med Genet C Semin Med Genet 139C: 4–9. - PubMed
-
- Freed LA, Levy D, Levine RA, Larson MG, Evans JC, et al. (1999) Prevalence and clinical outcome of mitral-valve prolapse. N Engl J Med 341: 1–7. - PubMed
-
- Judge DP, Dietz HC (2008) Therapy of Marfan syndrome. Annu Rev Med 59: 43–59. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

