Apigenin induces DNA damage through the PKCδ-dependent activation of ATM and H2AX causing down-regulation of genes involved in cell cycle control and DNA repair
- PMID: 22985621
- PMCID: PMC4097023
- DOI: 10.1016/j.bcp.2012.09.005
Apigenin induces DNA damage through the PKCδ-dependent activation of ATM and H2AX causing down-regulation of genes involved in cell cycle control and DNA repair
Abstract
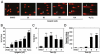
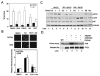
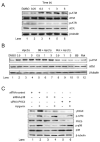
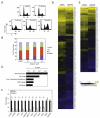
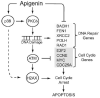
Apigenin, an abundant plant flavonoid, exhibits anti-proliferative and anti-carcinogenic activities through mechanisms yet not fully defined. In the present study, we show that the treatment of leukemia cells with apigenin resulted in the induction of DNA damage preceding the activation of the apoptotic program. Apigenin-induced DNA damage was mediated by p38 and protein kinase C-delta (PKCδ), yet was independent of reactive oxygen species or caspase activity. Treatment of monocytic leukemia cells with apigenin induced the phosphorylation of the ataxia-telangiectasia mutated (ATM) kinase and histone H2AX, two key regulators of the DNA damage response, without affecting the ataxia-telangiectasia mutated and Rad-3-related (ATR) kinase. Silencing and pharmacological inhibition of PKCδ abrogated ATM and H2AX phosphorylation, whereas inhibition of p38 reduced H2AX phosphorylation independently of ATM. We established that apigenin delayed cell cycle progression at G1/S and increased the number of apoptotic cells. In addition, genome-wide mRNA analyses showed that apigenin-induced DNA damage led to down-regulation of genes involved in cell-cycle control and DNA repair. Taken together, the present results show that the PKCδ-dependent activation of ATM and H2AX define the signaling networks responsible for the regulation of DNA damage promoting genome-wide mRNA alterations that result in cell cycle arrest, hence contributing to the anti-carcinogenic activities of this flavonoid.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Effects of hydroxyurea and aphidicolin on phosphorylation of ataxia telangiectasia mutated on Ser 1981 and histone H2AX on Ser 139 in relation to cell cycle phase and induction of apoptosis.Cytometry A. 2006 Apr;69(4):212-21. doi: 10.1002/cyto.a.20241. Cytometry A. 2006. PMID: 16528719
-
ATM activation accompanies histone H2AX phosphorylation in A549 cells upon exposure to tobacco smoke.BMC Cell Biol. 2007 Jun 26;8:26. doi: 10.1186/1471-2121-8-26. BMC Cell Biol. 2007. PMID: 17594478 Free PMC article.
-
DNA-PKcs plays a dominant role in the regulation of H2AX phosphorylation in response to DNA damage and cell cycle progression.BMC Mol Biol. 2010 Mar 6;11:18. doi: 10.1186/1471-2199-11-18. BMC Mol Biol. 2010. PMID: 20205745 Free PMC article.
-
Cytometry of ATM activation and histone H2AX phosphorylation to estimate extent of DNA damage induced by exogenous agents.Cytometry A. 2007 Sep;71(9):648-61. doi: 10.1002/cyto.a.20426. Cytometry A. 2007. PMID: 17622968 Free PMC article. Review.
-
Constitutive histone H2AX phosphorylation and ATM activation, the reporters of DNA damage by endogenous oxidants.Cell Cycle. 2006 Sep;5(17):1940-5. doi: 10.4161/cc.5.17.3191. Epub 2006 Sep 1. Cell Cycle. 2006. PMID: 16940754 Free PMC article. Review.
Cited by
-
Multifunctional roles of PKCδ: Opportunities for targeted therapy in human disease.Pharmacol Ther. 2016 Sep;165:1-13. doi: 10.1016/j.pharmthera.2016.05.001. Epub 2016 May 11. Pharmacol Ther. 2016. PMID: 27179744 Free PMC article. Review.
-
Flavones: From Biosynthesis to Health Benefits.Plants (Basel). 2016 Jun 21;5(2):27. doi: 10.3390/plants5020027. Plants (Basel). 2016. PMID: 27338492 Free PMC article. Review.
-
Dietary compounds have potential in controlling atherosclerosis by modulating macrophage cholesterol metabolism and inflammation via miRNA.NPJ Sci Food. 2018 Jul 25;2:13. doi: 10.1038/s41538-018-0022-8. eCollection 2018. NPJ Sci Food. 2018. PMID: 31304263 Free PMC article. Review.
-
Apigenin and hesperidin augment the toxic effect of doxorubicin against HepG2 cells.BMC Pharmacol Toxicol. 2019 May 3;20(1):22. doi: 10.1186/s40360-019-0301-2. BMC Pharmacol Toxicol. 2019. PMID: 31053173 Free PMC article.
-
Flavonoids: New Frontier for Immuno-Regulation and Breast Cancer Control.Antioxidants (Basel). 2019 Apr 16;8(4):103. doi: 10.3390/antiox8040103. Antioxidants (Basel). 2019. PMID: 30995775 Free PMC article. Review.
References
-
- Ross JA, Kasum CM. Dietary flavonoids: bioavailability, metabolic effects, and safety. Annu Rev Nutr. 2002;22:19–34. - PubMed
-
- Middleton E, Jr, Kandaswami C, Theoharides TC. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacol Rev. 2000;52:673–751. - PubMed
-
- Rice-Evans C, Spencer JP, Schroeter H, Rechner AR. Bioavailability of flavonoids and potential bioactive forms in vivo. Drug Metab Drug Interact. 2000;17:291–310. - PubMed
-
- Havsteen BH. The biochemistry and medical significance of the flavonoids. Pharmacol Ther. 2002;96:67–202. - PubMed
-
- Nielsen SE, Young JF, Daneshvar B, Lauridsen ST, Knuthsen P, Sandstrom B, et al. Effect of parsley (Petroselinum crispum) intake on urinary apigenin excretion, blood antioxidant enzymes and biomarkers for oxidative stress in human subjects. Br J Nutr. 1999;81:447–55. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

