Protective effects of alanyl-glutamine supplementation against nelfinavir-induced epithelial impairment in IEC-6 cells and in mouse intestinal mucosa
- PMID: 22986234
- PMCID: PMC3542240
- DOI: 10.4161/cbt.22251
Protective effects of alanyl-glutamine supplementation against nelfinavir-induced epithelial impairment in IEC-6 cells and in mouse intestinal mucosa
Abstract
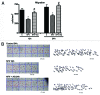
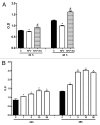
Purpose: Human Immunodeficiency Virus (HIV) protease inhibitors (PI) remain a crucial component of highly active therapy (HAART) and recently have been demonstrated to have potent antitumor effect on a wide variety of tumor cell lines. However, discontinuation of therapy is an important issue, which may be related to various side-effects, especially diarrhea. The aim of this study was to evaluate the effects of nelfinavir (NFV), an HIV PI, and of alanyl-glutamine (AQ) supplementation, on intestinal cell migration, proliferation, apoptosis and necrosis, using IEC-6 cells and on intestinal crypt depth, villus length, villus area, mitotic index and apoptosis in Swiss mice.
Methods: Migration was evaluated at 12 and 24 h after injury using a wound healing assay. Cellular proliferation was measured indirectly at 24 and 48 h using tetrazolium salt WST-1. Apoptosis and necrosis were measured by flow cytometry using the Annexin V assay. Intestinal morphometry and mitotic index in vivo were assessed following a seven-day treatment with 100 mg/kg of NFV, given orally. In vivo proliferation and apoptosis were evaluated by intestinal crypt mitotic index and immunohistochemistry, respectively.
Results: In vitro, AQ supplementation enhanced IEC-6 cell migration and proliferation, following challenge with NFV. In vivo, AQ increased intestinal villus length, villus area, crypt depth and cell proliferation and cell migration, following treatment with NFV. AQ did not decrease cell death induced by NFV both in vivo and in vitro.
Conclusions: AQ supplementation is potentially beneficial in preventing the effects of PIs, such as NFV, in the intestinal tract.
Figures



Similar articles
-
Evaluation of HIV protease and nucleoside reverse transcriptase inhibitors on proliferation, necrosis, apoptosis in intestinal epithelial cells and electrolyte and water transport and epithelial barrier function in mice.BMC Gastroenterol. 2010 Aug 11;10:90. doi: 10.1186/1471-230X-10-90. BMC Gastroenterol. 2010. PMID: 20701796 Free PMC article.
-
Intestinal epithelial restitution after TcdB challenge and recovery from Clostridium difficile infection in mice with alanyl-glutamine treatment.J Infect Dis. 2013 May 15;207(10):1505-15. doi: 10.1093/infdis/jit041. Epub 2013 Jan 28. J Infect Dis. 2013. PMID: 23359592 Free PMC article.
-
Alanyl-glutamine and glutamine supplementation improves 5-fluorouracil-induced intestinal epithelium damage in vitro.Dig Dis Sci. 2008 Oct;53(10):2687-96. doi: 10.1007/s10620-008-0215-0. Epub 2008 Mar 6. Dig Dis Sci. 2008. PMID: 18320312 Free PMC article.
-
Glucagon-like peptide 2 improves intestinal wound healing through induction of epithelial cell migration in vitro-evidence for a TGF--beta-mediated effect.Regul Pept. 2004 Sep 15;121(1-3):137-43. doi: 10.1016/j.regpep.2004.04.014. Regul Pept. 2004. PMID: 15256284
-
Antitumorigenic action of nelfinavir: Effects on multiple myeloma and hematologic malignancies (Review).Oncol Rep. 2020 Jun;43(6):1729-1736. doi: 10.3892/or.2020.7562. Epub 2020 Mar 26. Oncol Rep. 2020. PMID: 32236596 Review.
Cited by
-
Co-administration of Favipiravir and the Remdesivir Metabolite GS-441524 Effectively Reduces SARS-CoV-2 Replication in the Lungs of the Syrian Hamster Model.mBio. 2021 Feb 22;13(1):e0304421. doi: 10.1128/mbio.03044-21. Epub 2022 Feb 1. mBio. 2021. PMID: 35100870 Free PMC article.
-
Protection of rat liver against hepatic ischemia-reperfusion injury by a novel selenocysteine-containing 7-mer peptide.Mol Med Rep. 2016 Sep;14(3):2007-15. doi: 10.3892/mmr.2016.5464. Epub 2016 Jul 1. Mol Med Rep. 2016. PMID: 27431272 Free PMC article.
-
Effects of alanyl-glutamine supplementation on the small intestinal mucosa barrier in weaned piglets.Asian-Australas J Anim Sci. 2017 Feb;30(2):236-245. doi: 10.5713/ajas.16.0077. Epub 2016 Jun 30. Asian-Australas J Anim Sci. 2017. PMID: 27383799 Free PMC article.
-
Identification and characterization of PEDV infection in rat crypt epithelial cells.Vet Microbiol. 2020 Oct;249:108848. doi: 10.1016/j.vetmic.2020.108848. Epub 2020 Sep 17. Vet Microbiol. 2020. PMID: 32979749 Free PMC article.
References
-
- Bode H, Lenzner L, Kraemer OH, Kroesen AJ, Bendfeldt K, Schulzke JD, et al. The HIV protease inhibitors saquinavir, ritonavir, and nelfinavir induce apoptosis and decrease barrier function in human intestinal epithelial cells. Antivir Ther. 2005;10:645–55. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
