Genetic labeling of steroidogenic factor-1 (SF-1) neurons in mice reveals ventromedial nucleus of the hypothalamus (VMH) circuitry beginning at neurogenesis and development of a separate non-SF-1 neuronal cluster in the ventrolateral VMH
- PMID: 22987798
- PMCID: PMC4304766
- DOI: 10.1002/cne.23226
Genetic labeling of steroidogenic factor-1 (SF-1) neurons in mice reveals ventromedial nucleus of the hypothalamus (VMH) circuitry beginning at neurogenesis and development of a separate non-SF-1 neuronal cluster in the ventrolateral VMH
Abstract
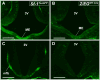
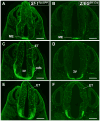
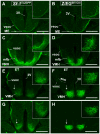
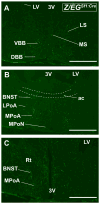
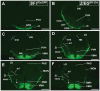
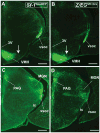
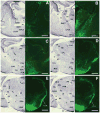
The ventromedial nucleus of the hypothalamus (VMH) influences a wide variety of physiological responses. Here, using two distinct but complementary genetic tracing approaches in mice, we describe the development of VMH efferent projections, as marked by steroidogenic factor-1 (SF-1; NR5A1). SF-1 neurons were visualized by Tau-green fluorescent protein (GFP) expressed from the endogenous Sf-1 locus (Sf-1(TauGFP)) or by crossing the transgenic Sf1:Cre driver to a GFP reporter strain (Z/EG(Sf1:Cre)). Strikingly, VMH projections were visible early, at embryonic (E) 10.5, when few postmitotic SF1 neurons have been born, suggesting that formation of VMH circuitry begins at the onset of neurogenesis. At E14.5, comparison of these two reporter lines revealed that SF1-positive neurons in the ventrolateral VMH (VMH(vl)) persist in Z/EG(Sf1:Cre) embryos but are virtually absent in Sf-1(TauGFP). Therefore, although the entire VMH including the VMH(vl) shares a common lineage, the VMH(vl) further differentiates into a neuronal cluster devoid of SF-1. At birth, extensive VMH projections to broad regions of the brain were observed in both mouse reporter lines, matching well with those previously discovered by injection of axonal anterograde tracers in adult rats. In summary, our genetic tracing studies show that VMH efferent projections are highly conserved in rodents and are established far earlier than previously appreciated. Moreover, our results imply that neurons in the VMH(vl) adopt a distinct fate early in development, which might underlie the unique physiological functions associated with this VMH subregion.
Copyright © 2012 Wiley Periodicals, Inc.
Conflict of interest statement
The authors have no conflict of interest and nothing to disclose.
Figures




References
-
- Cameron NM, Soehngen E, Meaney MJ. Variation in maternal care influences ventromedial hypothalamus activation in the rat. J Neuroendocrinol. 2011;23:393–400. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

