Metazoan Hsp70 machines use Hsp110 to power protein disaggregation
- PMID: 22990239
- PMCID: PMC3492728
- DOI: 10.1038/emboj.2012.264
Metazoan Hsp70 machines use Hsp110 to power protein disaggregation
Abstract
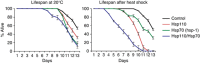
Accumulation of aggregation-prone misfolded proteins disrupts normal cellular function and promotes ageing and disease. Bacteria, fungi and plants counteract this by solubilizing and refolding aggregated proteins via a powerful cytosolic ATP-dependent bichaperone system, comprising the AAA+ disaggregase Hsp100 and the Hsp70-Hsp40 system. Metazoa, however, lack Hsp100 disaggregases. We show that instead the Hsp110 member of the Hsp70 superfamily remodels the human Hsp70-Hsp40 system to efficiently disaggregate and refold aggregates of heat and chemically denatured proteins in vitro and in cell extracts. This Hsp110 effect relies on nucleotide exchange, not on ATPase activity, implying ATP-driven chaperoning is not required. Knock-down of nematode Caenorhabditis elegans Hsp110, but not an unrelated nucleotide exchange factor, compromises dissolution of heat-induced protein aggregates and severely shortens lifespan after heat shock. We conclude that in metazoa, Hsp70-Hsp40 powered by Hsp110 nucleotide exchange represents the crucial disaggregation machinery that reestablishes protein homeostasis to counteract protein unfolding stress.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

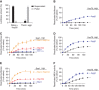
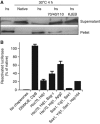
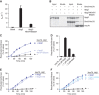

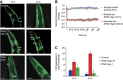
References
-
- Ali JA, Jackson AP, Howells AJ, Maxwell A (1993) The 43-kilodalton N-terminal fragment of the DNA gyrase B protein hydrolyzes ATP and binds coumarin drugs. Biochemistry 32: 2717–2724 - PubMed
-
- Andreasson C, Fiaux J, Rampelt H, Mayer MP, Bukau B (2008b) Hsp110 is a nucleotide-activated exchange factor for Hsp70. J Biol Chem 283: 8877–8884 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

