Shrinkage in nonlinear mixed-effects population models: quantification, influencing factors, and impact
- PMID: 22993107
- PMCID: PMC3475840
- DOI: 10.1208/s12248-012-9407-9
Shrinkage in nonlinear mixed-effects population models: quantification, influencing factors, and impact
Abstract
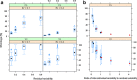
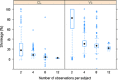
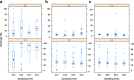
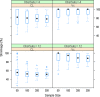
Shrinkage of empirical Bayes estimates (EBEs) of posterior individual parameters in mixed-effects models has been shown to obscure the apparent correlations among random effects and relationships between random effects and covariates. Empirical quantification equations have been widely used for population pharmacokinetic/pharmacodynamic models. The objectives of this manuscript were (1) to compare the empirical equations with theoretically derived equations, (2) to investigate and confirm the influencing factor on shrinkage, and (3) to evaluate the impact of shrinkage on estimation errors of EBEs using Monte Carlo simulations. A mathematical derivation was first provided for the shrinkage in nonlinear mixed effects model. Using a linear mixed model, the simulation results demonstrated that the shrinkage estimated from the empirical equations matched those based on the theoretically derived equations. Simulations with a two-compartment pharmacokinetic model verified that shrinkage has a reversed relationship with the relative ratio of interindividual variability to residual variability. Fewer numbers of observations per subject were associated with higher amount of shrinkage, consistent with findings from previous research. The influence of sampling times appeared to be larger when fewer PK samples were collected for each individual. As expected, sample size has very limited impact on shrinkage of the PK parameters of the two-compartment model. Assessment of estimation error suggested an average 1:1 relationship between shrinkage and median estimation error of EBEs.
Figures


Similar articles
-
Further Evaluation of Covariate Analysis using Empirical Bayes Estimates in Population Pharmacokinetics: the Perception of Shrinkage and Likelihood Ratio Test.AAPS J. 2017 Jan;19(1):264-273. doi: 10.1208/s12248-016-0001-4. Epub 2016 Oct 19. AAPS J. 2017. PMID: 27761720
-
Prediction of shrinkage of individual parameters using the bayesian information matrix in non-linear mixed effect models with evaluation in pharmacokinetics.Pharm Res. 2013 Sep;30(9):2355-67. doi: 10.1007/s11095-013-1079-3. Epub 2013 Jun 7. Pharm Res. 2013. PMID: 23743656
-
Importance of shrinkage in empirical bayes estimates for diagnostics: problems and solutions.AAPS J. 2009 Sep;11(3):558-69. doi: 10.1208/s12248-009-9133-0. Epub 2009 Aug 1. AAPS J. 2009. PMID: 19649712 Free PMC article.
-
Pharmacokinetic comparability between two populations using nonlinear mixed effect models: a Monte Carlo study.J Pharmacokinet Pharmacodyn. 2023 Jun;50(3):189-201. doi: 10.1007/s10928-023-09842-2. Epub 2023 Jan 28. J Pharmacokinet Pharmacodyn. 2023. PMID: 36708443
-
Investigating the relationship between the Bayes factor and the separation of credible intervals.Psychon Bull Rev. 2023 Oct;30(5):1759-1781. doi: 10.3758/s13423-023-02295-1. Epub 2023 May 11. Psychon Bull Rev. 2023. PMID: 37170004 Review.
Cited by
-
Impact of Sampling Period on Population Pharmacokinetic Analysis of Antibiotics: Why do You Take Blood Samples Following the Fourth Dose?Pharmaceuticals (Basel). 2020 Sep 16;13(9):249. doi: 10.3390/ph13090249. Pharmaceuticals (Basel). 2020. PMID: 32947890 Free PMC article.
-
Powers of the likelihood ratio test and the correlation test using empirical bayes estimates for various shrinkages in population pharmacokinetics.CPT Pharmacometrics Syst Pharmacol. 2014 Apr 9;3(4):e109. doi: 10.1038/psp.2014.5. CPT Pharmacometrics Syst Pharmacol. 2014. PMID: 24717242 Free PMC article.
-
"De-Shrinking" EBEs: The Solution for Bayesian Therapeutic Drug Monitoring.Clin Pharmacokinet. 2022 May;61(5):749-757. doi: 10.1007/s40262-021-01105-y. Epub 2022 Feb 4. Clin Pharmacokinet. 2022. PMID: 35119624 Free PMC article.
-
Effect of Renal Impairment on the Pharmacokinetics and Safety of Axitinib.Target Oncol. 2016 Apr;11(2):229-34. doi: 10.1007/s11523-015-0389-2. Target Oncol. 2016. PMID: 26400730 Free PMC article.
-
Toward Model-Based Informed Precision Dosing of Vancomycin in Hematologic Cancer Patients: A First Step.Clin Pharmacokinet. 2024 Feb;63(2):183-196. doi: 10.1007/s40262-023-01329-0. Epub 2023 Dec 21. Clin Pharmacokinet. 2024. PMID: 38127240
References
-
- Fitzmaurice GM. Longitudinal data analysis. Boca Raton: Chapman & Hall; 2008.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

