Dynamics of the establishment of systemic Potyvirus infection: independent yet cumulative action of primary infection sites
- PMID: 22993154
- PMCID: PMC3497618
- DOI: 10.1128/JVI.02207-12
Dynamics of the establishment of systemic Potyvirus infection: independent yet cumulative action of primary infection sites
Abstract
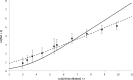
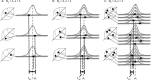
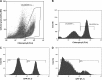
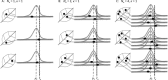
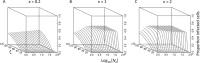
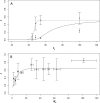
In the clinic, farm, or field, for many viruses there is a high prevalence of mixed-genotype infections, indicating that multiple virions have initiated infection and that there can be multiple sites of primary infection within the same host. The dynamic process by which multiple primary infection sites interact with each other and the host is poorly understood, undoubtedly due to its high complexity. In this study, we attempted to unravel the basic interactions underlying this process using a plant RNA virus, as removing the inoculated leaf can instantly and rigorously eliminate all primary infection sites. Effective population size in the inoculated leaf and time of removal of the inoculated leaf were varied in experiments, and it was found that both factors positively influenced if the plant became systemically infected and what proportion of cells in the systemic tissue were infected, as measured by flow cytometry. Fitting of probabilistic models of infection to our data demonstrated that a null model in which the action of each focus is independent of the presence of other foci was better supported than a dependent-action model. The cumulative effect of independently acting foci therefore determined when plants became infected and how many individual cells were infected. There was no evidence for interference between primary infection sites, which is surprising given the planar structure of leaves. By showing that a simple null model is supported, we experimentally confirmed--to our knowledge for the first time--the minimal components that dictate interactions of a conspecific virus population establishing systemic infection.
Figures


Similar articles
-
One is enough: in vivo effective population size is dose-dependent for a plant RNA virus.PLoS Pathog. 2011 Jul;7(7):e1002122. doi: 10.1371/journal.ppat.1002122. Epub 2011 Jul 7. PLoS Pathog. 2011. PMID: 21750676 Free PMC article.
-
Effects of potyvirus effective population size in inoculated leaves on viral accumulation and the onset of symptoms.J Virol. 2012 Sep;86(18):9737-47. doi: 10.1128/JVI.00909-12. Epub 2012 Jun 27. J Virol. 2012. PMID: 22740417 Free PMC article.
-
Potyvirus-induced gene silencing: the dynamic process of systemic silencing and silencing suppression.J Gen Virol. 2007 Aug;88(Pt 8):2337-2346. doi: 10.1099/vir.0.82928-0. J Gen Virol. 2007. PMID: 17622640
-
Infection dynamics in viral spread and interference under the synergism between Cucumber mosaic virus and Turnip mosaic virus.Mol Plant Microbe Interact. 2012 Jan;25(1):18-27. doi: 10.1094/MPMI-06-11-0170. Mol Plant Microbe Interact. 2012. PMID: 21916556
-
Constraints to virus infection in Nicotiana benthamiana plants transformed with a potyvirus amplicon.BMC Plant Biol. 2010 Jul 6;10:139. doi: 10.1186/1471-2229-10-139. BMC Plant Biol. 2010. PMID: 20604920 Free PMC article.
Cited by
-
Model-selection-based approach for calculating cellular multiplicity of infection during virus colonization of multi-cellular hosts.PLoS One. 2013 May 28;8(5):e64657. doi: 10.1371/journal.pone.0064657. Print 2013. PLoS One. 2013. PMID: 23724074 Free PMC article.
-
Narrow bottlenecks affect Pea seedborne mosaic virus populations during vertical seed transmission but not during leaf colonization.PLoS Pathog. 2014 Jan;10(1):e1003833. doi: 10.1371/journal.ppat.1003833. Epub 2014 Jan 9. PLoS Pathog. 2014. PMID: 24415934 Free PMC article.
-
Impact of genetic drift, selection and accumulation level on virus adaptation to its host plants.Mol Plant Pathol. 2018 Dec;19(12):2575-2589. doi: 10.1111/mpp.12730. Epub 2018 Oct 25. Mol Plant Pathol. 2018. PMID: 30074299 Free PMC article.
-
Significantly Improved Recovery of Recombinant Sonchus Yellow Net Rhabdovirus by Expressing the Negative-Strand Genomic RNA.Viruses. 2020 Dec 17;12(12):1459. doi: 10.3390/v12121459. Viruses. 2020. PMID: 33348798 Free PMC article.
-
Transcriptional and hormonal profiling uncovers the interactions between plant developmental stages and RNA virus infection.J Gen Virol. 2024 Sep;105(9):002023. doi: 10.1099/jgv.0.002023. J Gen Virol. 2024. PMID: 39292505
References
-
- Bald JG. 1937. The use of numbers of infections for comparing the concentration of plant virus suspensions. I. Dilution experiments with purified suspensions. Ann. Appl. Biol. 24:33–55
-
- Carrasco P, Daròs JA, Agudelo-Romero P, Elena SF. 2007. A real-time RT-PCR assay for quantifying the fitness of Tobacco etch virus in competition experiments. J. Virol. Methods 139:181–188 - PubMed

