A Tetrapyrrole Macrocycle Displaying a Multielectron Redox Chemistry and Tunable Absorbance Profile
- PMID: 22993642
- PMCID: PMC3443682
- DOI: 10.1021/jp3059382
A Tetrapyrrole Macrocycle Displaying a Multielectron Redox Chemistry and Tunable Absorbance Profile
Abstract
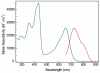
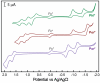
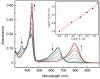
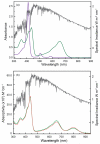
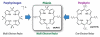
Porphyrins are attractive chromophores for incorporation into light harvesting devices. Some of the most efficient porphyrin derivatives in this regard are synthetically complex platforms with specially tailored electronic properties. This work details the unique geometric and electronic structure of the phlorin framework. X-ray crystallography and NMR spectroscopy demonstrate that unlike typical tetrapyrrole macrocycles, the phlorin is not aromatic. These unusual electronics are manifest in distinct photophysical and redox properties, as the phlorin displays a rich multielectron redox chemistry. The phlorin also displays an intriguing supramolecular chemistry and can reversibly bind up to two equivalents of fluoride in cooperative fashion. Accordingly, this synthetically accessible sensitizer displays a rich multielectron redox chemistry, excellent spectral coverage and an intriguing anion binding chemistry that distinguishes this system from more commonly studied porphyrinoids.
Figures
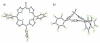
Similar articles
-
Synthesis, electrochemistry, and photophysics of a family of phlorin macrocycles that display cooperative fluoride binding.J Am Chem Soc. 2013 May 1;135(17):6601-7. doi: 10.1021/ja401391z. Epub 2013 Apr 17. J Am Chem Soc. 2013. PMID: 23594346 Free PMC article.
-
Factors Controlling the Spectroscopic Properties and Supramolecular Chemistry of an Electron Deficient 5,5-Dimethylphlorin Architecture.J Phys Chem C Nanomater Interfaces. 2014 Jul 3;118(26):14124-14132. doi: 10.1021/jp5016824. Epub 2014 May 29. J Phys Chem C Nanomater Interfaces. 2014. PMID: 25018789 Free PMC article.
-
Porpholactone Chemistry: An Emerging Approach to Bioinspired Photosensitizers with Tunable Near-Infrared Photophysical Properties.Acc Chem Res. 2019 Sep 17;52(9):2620-2633. doi: 10.1021/acs.accounts.9b00119. Epub 2019 Jul 12. Acc Chem Res. 2019. PMID: 31298833 Review.
-
Synthesis of Phlorin Analogues of Dithiacorrphycene and Their Use as Specific Chemodosimetric Sensors for Fe3+ Ions.Chem Asian J. 2018 Oct 18;13(20):3040-3050. doi: 10.1002/asia.201801005. Epub 2018 Sep 19. Chem Asian J. 2018. PMID: 30009561
-
Bioinspired Applications of Porphyrin Derivatives.Acc Chem Res. 2021 May 4;54(9):2249-2260. doi: 10.1021/acs.accounts.1c00114. Epub 2021 Apr 23. Acc Chem Res. 2021. PMID: 33891405 Review.
Cited by
-
Synthesis, Redox, and Spectroscopic Properties of Pd(II) 10,10-Dimethylisocorrole Complexes Prepared via Bromination of Dimethylbiladiene Oligotetrapyrroles.Inorg Chem. 2020 Dec 21;59(24):18241-18252. doi: 10.1021/acs.inorgchem.0c02721. Epub 2020 Dec 7. Inorg Chem. 2020. PMID: 33284618 Free PMC article.
-
Synthesis, electrochemistry, and photophysics of a family of phlorin macrocycles that display cooperative fluoride binding.J Am Chem Soc. 2013 May 1;135(17):6601-7. doi: 10.1021/ja401391z. Epub 2013 Apr 17. J Am Chem Soc. 2013. PMID: 23594346 Free PMC article.
-
Mapping the influence of ligand electronics on the spectroscopic and 1O2 sensitization characteristics of Pd(II) biladiene complexes bearing phenyl-alkynyl groups at the 2- and 18-positions.Dalton Trans. 2023 Jun 6;52(22):7512-7523. doi: 10.1039/d3dt00691c. Dalton Trans. 2023. PMID: 37199710 Free PMC article.
-
The Eponymous Cofactors in Cytochrome P460s from Ammonia-Oxidizing Bacteria Are Iron Porphyrinoids Whose Macrocycles Are Dibasic.Biochemistry. 2018 Jan 23;57(3):334-343. doi: 10.1021/acs.biochem.7b00921. Epub 2017 Dec 6. Biochemistry. 2018. PMID: 29211462 Free PMC article.
-
Reversible π-system switching of thiophene-fused thiahexaphyrins by solvent and oxidation/reduction.Chem Sci. 2018 Aug 14;9(38):7528-7539. doi: 10.1039/c8sc02448k. eCollection 2018 Oct 14. Chem Sci. 2018. PMID: 30319753 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
