Sublingual immunization with recombinant adenovirus encoding SARS-CoV spike protein induces systemic and mucosal immunity without redirection of the virus to the brain
- PMID: 22995185
- PMCID: PMC3489719
- DOI: 10.1186/1743-422X-9-215
Sublingual immunization with recombinant adenovirus encoding SARS-CoV spike protein induces systemic and mucosal immunity without redirection of the virus to the brain
Abstract
Background: Sublingual (s.l.) administration of soluble protein antigens, inactivated viruses, or virus-like particles has been shown to induce broad immune responses in mucosal and extra-mucosal tissues. Recombinant replication-defective adenovirus vectors (rADVs) infect mucosa surface and therefore can serve as a mucosal antigen delivery vehicle. In this study we examined whether s.l. immunization with rADV encoding spike protein (S) (rADV-S) of severe acute respiratory syndrome-associated coronavirus (SARS-CoV) induces protective immunity against SARS-CoV and could serve as a safe mucosal route for delivery of rADV.
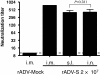
Results: Here, we show that s.l. administration of rADV-S induced serum SARS-CoV neutralizing and airway IgA antibodies in mice. These antibody responses are comparable to those induced by intranasal (i.n.) administration. In addition, s.l. immunization induced antigen-specific CD8+ T cell responses in the lungs that are superior to those induced by intramuscular immunization. Importantly, unlike i.n. administration, s.l. immunization with rADV did not redirect the rADV vector to the olfactory bulb.
Conclusion: Our study indicates that s.l. immunization with rADV-S is safe and effective in induction of a broad spectrum of immune responses and presumably protection against infection with SARS-CoV.
Figures
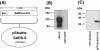
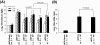
Similar articles
-
Intranasal vaccination of recombinant adeno-associated virus encoding receptor-binding domain of severe acute respiratory syndrome coronavirus (SARS-CoV) spike protein induces strong mucosal immune responses and provides long-term protection against SARS-CoV infection.J Immunol. 2008 Jan 15;180(2):948-56. doi: 10.4049/jimmunol.180.2.948. J Immunol. 2008. PMID: 18178835 Free PMC article.
-
Contributions of the structural proteins of severe acute respiratory syndrome coronavirus to protective immunity.Proc Natl Acad Sci U S A. 2004 Jun 29;101(26):9804-9. doi: 10.1073/pnas.0403492101. Epub 2004 Jun 21. Proc Natl Acad Sci U S A. 2004. PMID: 15210961 Free PMC article.
-
Comparative evaluation of two severe acute respiratory syndrome (SARS) vaccine candidates in mice challenged with SARS coronavirus.J Gen Virol. 2006 Mar;87(Pt 3):641-650. doi: 10.1099/vir.0.81579-0. J Gen Virol. 2006. PMID: 16476986
-
Vaccine design for severe acute respiratory syndrome coronavirus.Viral Immunol. 2005;18(2):327-32. doi: 10.1089/vim.2005.18.327. Viral Immunol. 2005. PMID: 16035944 Review.
-
SARS Immunity and Vaccination.Cell Mol Immunol. 2004 Jun;1(3):193-8. Cell Mol Immunol. 2004. PMID: 16219167 Review.
Cited by
-
Immune Response, Inflammation, and the Clinical Spectrum of COVID-19.Front Immunol. 2020 Jun 16;11:1441. doi: 10.3389/fimmu.2020.01441. eCollection 2020. Front Immunol. 2020. PMID: 32612615 Free PMC article. Review.
-
Sublingual delivery of vaccines for the induction of mucosal immunity.Immune Netw. 2013 Jun;13(3):81-5. doi: 10.4110/in.2013.13.3.81. Epub 2013 Jun 30. Immune Netw. 2013. PMID: 23885221 Free PMC article.
-
Severe acute respiratory syndrome-coronavirus-2 spike (S) protein based vaccine candidates: State of the art and future prospects.Rev Med Virol. 2021 May;31(3):e2183. doi: 10.1002/rmv.2183. Epub 2020 Oct 15. Rev Med Virol. 2021. PMID: 33594794 Free PMC article. Review.
-
An Old Acquaintance: Could Adenoviruses Be Our Next Pandemic Threat?Viruses. 2023 Jan 24;15(2):330. doi: 10.3390/v15020330. Viruses. 2023. PMID: 36851544 Free PMC article. Review.
-
Microneedle-Mediated Vaccine Delivery to the Oral Mucosa.Adv Healthc Mater. 2019 Feb;8(4):e1801180. doi: 10.1002/adhm.201801180. Epub 2018 Dec 10. Adv Healthc Mater. 2019. PMID: 30537400 Free PMC article. Review.
References
-
- Osek J, Truszczynski M. [Mucosal immunity with implications for use in developing a new generation of vaccines] Postepy Hig Med Dosw. 1995;49:469–486. - PubMed
-
- See R, Zakhartchouk A, Petric M, Lawrence D, Mok C, Hogan R, Rowe T, Zitzow L, Karunakaran K, Hitt M. Comparative evaluation of two severe acute respiratory syndrome (SARS) vaccine candidates in mice challenged with SARS coronavirus. J Gen Virol. 2006;87:641–650. doi: 10.1099/vir.0.81579-0. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

