Cell mechanics, structure, and function are regulated by the stiffness of the three-dimensional microenvironment
- PMID: 22995491
- PMCID: PMC3446717
- DOI: 10.1016/j.bpj.2012.07.054
Cell mechanics, structure, and function are regulated by the stiffness of the three-dimensional microenvironment
Abstract
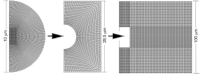
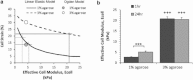
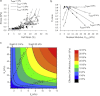
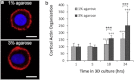
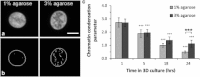
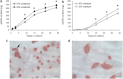
This study adopts a combined computational and experimental approach to determine the mechanical, structural, and metabolic properties of isolated chondrocytes cultured within three-dimensional hydrogels. A series of linear elastic and hyperelastic finite-element models demonstrated that chondrocytes cultured for 24 h in gels for which the relaxation modulus is <5 kPa exhibit a cellular Young's modulus of ∼5 kPa. This is notably greater than that reported for isolated chondrocytes in suspension. The increase in cell modulus occurs over a 24-h period and is associated with an increase in the organization of the cortical actin cytoskeleton, which is known to regulate cell mechanics. However, there was a reduction in chromatin condensation, suggesting that changes in the nucleus mechanics may not be involved. Comparison of cells in 1% and 3% agarose showed that cells in the stiffer gels rapidly develop a higher Young's modulus of ∼20 kPa, sixfold greater than that observed in the softer gels. This was associated with higher levels of actin organization and chromatin condensation, but only after 24 h in culture. Further studies revealed that cells in stiffer gels synthesize less extracellular matrix over a 28-day culture period. Hence, this study demonstrates that the properties of the three-dimensional microenvironment regulate the mechanical, structural, and metabolic properties of living cells.
Copyright © 2012 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Biomechanical properties of single chondrocytes and chondrons determined by micromanipulation and finite-element modelling.J R Soc Interface. 2010 Dec 6;7(53):1723-33. doi: 10.1098/rsif.2010.0207. Epub 2010 Jun 2. J R Soc Interface. 2010. PMID: 20519215 Free PMC article.
-
The effect of matrix stiffness on biomechanical properties of chondrocytes.Acta Biochim Biophys Sin (Shanghai). 2016 Oct;48(10):958-965. doi: 10.1093/abbs/gmw087. Epub 2016 Sep 2. Acta Biochim Biophys Sin (Shanghai). 2016. PMID: 27590061
-
Substrate stiffness together with soluble factors affects chondrocyte mechanoresponses.ACS Appl Mater Interfaces. 2014 Sep 24;6(18):16106-16. doi: 10.1021/am504135b. Epub 2014 Sep 5. ACS Appl Mater Interfaces. 2014. PMID: 25162787
-
The deformation behavior and mechanical properties of chondrocytes in articular cartilage.Osteoarthritis Cartilage. 1999 Jan;7(1):59-70. doi: 10.1053/joca.1998.0162. Osteoarthritis Cartilage. 1999. PMID: 10367015 Review.
-
Elasticity spectra as a tool to investigate actin cortex mechanics.J Nanobiotechnology. 2020 Oct 20;18(1):147. doi: 10.1186/s12951-020-00706-2. J Nanobiotechnology. 2020. PMID: 33081777 Free PMC article. Review.
Cited by
-
Relations between Structure/Composition and Mechanics in Osteoarthritic Regenerated Articular Tissue: A Machine Learning Approach.Int J Mol Sci. 2023 Aug 29;24(17):13374. doi: 10.3390/ijms241713374. Int J Mol Sci. 2023. PMID: 37686179 Free PMC article.
-
Nanofiber-microwell cell culture system for spatially patterned differentiation of pluripotent stem cells in 3D.Mater Today Bio. 2024 Jun 1;26:101109. doi: 10.1016/j.mtbio.2024.101109. eCollection 2024 Jun. Mater Today Bio. 2024. PMID: 38883422 Free PMC article.
-
Derivation of Mesenchymal Stem Cells through Sequential Presentation of Growth Factors via Gelatin Microparticles in Pluripotent Stem Cell Spheroids.Biomater Res. 2025 Apr 29;29:0184. doi: 10.34133/bmr.0184. eCollection 2025. Biomater Res. 2025. PMID: 40303481 Free PMC article.
-
Indenting at the Microscale: Guidelines for Robust Mechanical Characterization of Alginate Microgels.ACS Appl Mater Interfaces. 2025 Mar 5;17(9):13513-13526. doi: 10.1021/acsami.4c20952. Epub 2025 Feb 24. ACS Appl Mater Interfaces. 2025. PMID: 39994022 Free PMC article.
-
A-to-I RNA editing of Filamin A regulates cellular adhesion, migration and mechanical properties.FEBS J. 2022 Aug;289(15):4580-4601. doi: 10.1111/febs.16391. Epub 2022 Mar 9. FEBS J. 2022. PMID: 35124883 Free PMC article.
References
-
- Lee D.A., Knight M.M., Bader D.L. Stem cell mechanobiology. J. Cell. Biochem. 2011;112:1–9. - PubMed
-
- Ingber D.E., Dike L., Wang N. Cellular tensegrity: exploring how mechanical changes in the cytoskeleton regulate cell growth, migration, and tissue pattern during morphogenesis. Int. Rev. Cytol. 1994;150:173–224. - PubMed
-
- Ingber D.E. Mechanobiology and diseases of mechanotransduction. Ann. Med. 2003;35:564–577. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

