Non-amyloid and amyloid prion protein deposits in prion-infected mice differ in blockage of interstitial brain fluid
- PMID: 22998478
- PMCID: PMC3567241
- DOI: 10.1111/j.1365-2990.2012.01303.x
Non-amyloid and amyloid prion protein deposits in prion-infected mice differ in blockage of interstitial brain fluid
Abstract
Aims: Prion diseases are characterized by brain deposits of misfolded aggregated protease-resistant prion protein (PrP), termed PrPres. In humans and animals, PrPres is found as either disorganized non-amyloid aggregates or organized amyloid fibrils. Both PrPres forms are found in extracellular spaces of the brain. Thus, both might block drainage of brain interstitial fluid (ISF). The present experiments studied whether ISF blockage occurred during amyloid and/or non-amyloid prion diseases.
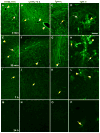
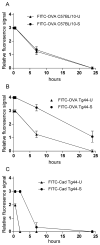
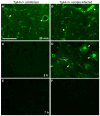
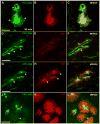
Methods: Various-sized fluorescein-labelled ISF tracers were stereotactically inoculated into the striatum of adult mice. At times from 5 min to 77 h, uninfected and scrapie-infected mice were compared. C57BL/10 mice expressing wild-type anchored PrP, which develop non-amyloid PrPres similar to humans with sporadic Creutzfeldt-Jakob disease, were compared with Tg44+/+ mice (transgenic mice secreting anchorless PrP) expressing anchorless PrP, which develop amyloid PrPres similar to certain human familial prion diseases.
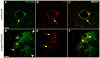
Results: In C57BL/10 mice, extensive non-amyloid PrPres aggregate deposition was not associated with abnormal clearance kinetics of tracers. In contrast, scrapie-infected Tg44+/+ mice showed blockage of tracer clearance and colocalization of tracer with perivascular PrPres amyloid.
Conclusions: As tracer localization and clearance was normal in infected C57BL/10 mice, ISF blockage was not an important pathogenic mechanism in this model. Therefore, ISF blockage is unlikely to be a problem in non-amyloid human prion diseases such as sporadic Creutzfeldt-Jakob disease. In contrast, partial ISF blockage appeared to be a possible pathogenic mechanism in Tg44+/+ mice. Thus this mechanism might also influence human amyloid prion diseases where expression of anchorless or mutated PrP results in perivascular amyloid PrPres deposition and cerebral amyloid angiopathy.
Published 2012. This article is a U.S. Government work and is in the public domain in the USA.
Figures

Similar articles
-
Distinct patterns of spread of prion infection in brains of mice expressing anchorless or anchored forms of prion protein.Acta Neuropathol Commun. 2014 Jan 21;2:8. doi: 10.1186/2051-5960-2-8. Acta Neuropathol Commun. 2014. PMID: 24447368 Free PMC article.
-
Unusual cerebral vascular prion protein amyloid distribution in scrapie-infected transgenic mice expressing anchorless prion protein.Acta Neuropathol Commun. 2013 Jun 19;1:25. doi: 10.1186/2051-5960-1-25. Acta Neuropathol Commun. 2013. PMID: 24252347 Free PMC article.
-
Fatal transmissible amyloid encephalopathy: a new type of prion disease associated with lack of prion protein membrane anchoring.PLoS Pathog. 2010 Mar 5;6(3):e1000800. doi: 10.1371/journal.ppat.1000800. PLoS Pathog. 2010. PMID: 20221436 Free PMC article.
-
The neuropathological phenotype in transgenic mice expressing different prion protein constructs.Philos Trans R Soc Lond B Biol Sci. 1994 Mar 29;343(1306):415-23. doi: 10.1098/rstb.1994.0038. Philos Trans R Soc Lond B Biol Sci. 1994. PMID: 7913760 Review.
-
[A process of programmed cell death as a mechanisms of neuronal death in prion diseases].Clin Exp Pathol. 1999;47(3-4):181-91. Clin Exp Pathol. 1999. PMID: 10472738 Review. French.
Cited by
-
Distinct patterns of spread of prion infection in brains of mice expressing anchorless or anchored forms of prion protein.Acta Neuropathol Commun. 2014 Jan 21;2:8. doi: 10.1186/2051-5960-2-8. Acta Neuropathol Commun. 2014. PMID: 24447368 Free PMC article.
-
PrP P102L and Nearby Lysine Mutations Promote Spontaneous In Vitro Formation of Transmissible Prions.J Virol. 2017 Oct 13;91(21):e01276-17. doi: 10.1128/JVI.01276-17. Print 2017 Nov 1. J Virol. 2017. PMID: 28835493 Free PMC article.
-
Noninvasive Antemortem Detection of Retinal Prions by a Fluorescent Tracer.J Alzheimers Dis. 2022;88(3):1137-1145. doi: 10.3233/JAD-220314. J Alzheimers Dis. 2022. PMID: 35754278 Free PMC article.
-
Probing early misfolding events in prion protein mutants by NMR spectroscopy.Molecules. 2013 Aug 7;18(8):9451-76. doi: 10.3390/molecules18089451. Molecules. 2013. PMID: 23966072 Free PMC article. Review.
-
Unusual cerebral vascular prion protein amyloid distribution in scrapie-infected transgenic mice expressing anchorless prion protein.Acta Neuropathol Commun. 2013 Jun 19;1:25. doi: 10.1186/2051-5960-1-25. Acta Neuropathol Commun. 2013. PMID: 24252347 Free PMC article.
References
-
- Parchi P, Giese A, Capellari S, Brown P, Schulz-Schaeffer W, Windl O, Zerr I, Budka H, Kopp N, Piccardo P, Poser S, Rojiani A, Streichemberger N, Julien J, Vital C, Ghetti B, Gambetti P, Kretzschmar H. Classification of sporadic Creutzfeldt-Jakob disease based on molecular and phenotypic analysis of 300 subjects. Ann Neurol. 1999;46:224–33. - PubMed
-
- Budka H, Aguzzi A, Brown P, Brucher JM, Bugiani O, Gullotta F, Haltia M, Hauw JJ, Ironside JW, Jellinger K, et al. Neuropathological diagnostic criteria for Creutzfeldt-Jakob disease (CJD) and other human spongiform encephalopathies (prion diseases) Brain Pathol. 1995;5:459–66. - PubMed
-
- Gonzalez L, Martin S, Begara-McGorum I, Hunter N, Houston F, Simmons M, Jeffrey M. Effects of agent strain and host genotype on PrP accumulation in the brain of sheep naturally and experimentally affected with scrapie. J Comp Pathol. 2002;126:17–29. - PubMed
-
- Jeffrey M, Goodsir CM, Bruce ME, McBride PA, Fraser JR. In vivo toxicity of prion protein in murine scrapie: ultrastructural and immunogold studies. Neuropathol Appl Neurobiol. 1997;23:93–101. - PubMed
-
- Ghetti B, Piccardo P, Frangione B, Bugiani O, Giaccone G, Young K, Prelli F, Farlow MR, Dlouhy SR, Tagliavini F. Prion protein amyloidosis. Brain Pathol. 1996;6:127–45. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

