Metabolic communication between astrocytes and neurons via bicarbonate-responsive soluble adenylyl cyclase
- PMID: 22998876
- PMCID: PMC3630998
- DOI: 10.1016/j.neuron.2012.08.032
Metabolic communication between astrocytes and neurons via bicarbonate-responsive soluble adenylyl cyclase
Abstract
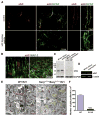
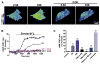
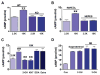
Astrocytes are proposed to participate in brain energy metabolism by supplying substrates to neurons from their glycogen stores and from glycolysis. However, the molecules involved in metabolic sensing and the molecular pathways responsible for metabolic coupling between different cell types in the brain are not fully understood. Here we show that a recently cloned bicarbonate (HCO₃⁻) sensor, soluble adenylyl cyclase (sAC), is highly expressed in astrocytes and becomes activated in response to HCO₃⁻ entry via the electrogenic NaHCO₃ cotransporter (NBC). Activated sAC increases intracellular cAMP levels, causing glycogen breakdown, enhanced glycolysis, and the release of lactate into the extracellular space, which is subsequently taken up by neurons for use as an energy substrate. This process is recruited over a broad physiological range of [K⁺](ext) and also during aglycemic episodes, helping to maintain synaptic function. These data reveal a molecular pathway in astrocytes that is responsible for brain metabolic coupling to neurons.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures




References
-
- Alle H, Roth A, Geiger JR. Energy-efficient action potentials in hippocampal mossy fibers. Science. 2009;325:1405–1408. - PubMed
-
- Attwell D, Laughlin SB. An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab. 2001;21:1133–1145. - PubMed
-
- Beierlein M, Gee KR, Martin VV, Regehr WG. Presynaptic calcium measurements at physiological temperatures using a new class of dextran-conjugated indicators. J Neurophysiol. 2004;92:591–599. - PubMed
-
- Bengtsson F, Boris-Möller F, Hansen AJ, Siesjö BK. Extracellular pH in the rat brain during hypoglycemic coma and recovery. J Cereb Blood Flow Metab. 1990;10:262–269. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

