The spatial structure of a nonlinear receptive field
- PMID: 23001060
- PMCID: PMC3517818
- DOI: 10.1038/nn.3225
The spatial structure of a nonlinear receptive field
Abstract
Understanding a sensory system implies the ability to predict responses to a variety of inputs from a common model. In the retina, this includes predicting how the integration of signals across visual space shapes the outputs of retinal ganglion cells. Existing models of this process generalize poorly to predict responses to new stimuli. This failure arises in part from properties of the ganglion cell response that are not well captured by standard receptive-field mapping techniques: nonlinear spatial integration and fine-scale heterogeneities in spatial sampling. Here we characterize a ganglion cell's spatial receptive field using a mechanistic model based on measurements of the physiological properties and connectivity of only the primary excitatory circuitry of the retina. The resulting simplified circuit model successfully predicts ganglion-cell responses to a variety of spatial patterns and thus provides a direct correspondence between circuit connectivity and retinal output.
Figures
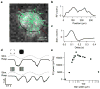

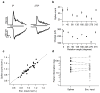
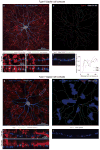
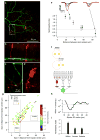
References
-
- Chichilnisky EJ. A simple white noise analysis of neuronal light responses. Network. 2001;12:199–213. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

