Immunoreactivities of androgen receptor, estrogen receptors, p450arom, p450c17 proteins in wild ground squirrels ovaries during the nonbreeding and breeding seasons
- PMID: 23009195
- PMCID: PMC3473255
- DOI: 10.1186/1757-2215-5-26
Immunoreactivities of androgen receptor, estrogen receptors, p450arom, p450c17 proteins in wild ground squirrels ovaries during the nonbreeding and breeding seasons
Abstract
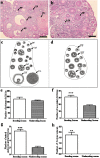
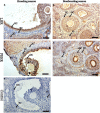
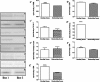
The aim of this study was to elucidate the regulatory role of androgen in the follicular development of wild female ground squirrels. Immunohistochemical staining of FSHR, LHR, P450c17, P450arom, androgen receptor (AR), estrogen receptors (ERa and ERb) were executed in ovaries of female ground squirrels from both breeding and nonbreeding seasons. In addition, total ovarian proteins were extracted from the ovaries of squirrels from breeding and nonbreeding seasons, and Western blot analysis were performed in order to probe for FSHR, LHR, P450c17, P450arom, AR, ERa and ERb. The results of immunohistochemical staining and Western blotting of P450c17 showed that there was no significant difference between the breeding and nonbreeding seasons. It was found that granulosa cells expressed P450arom during the breeding season. In contrast, there was no positive staining of P450arom in the nonbreeding season. There was no significant difference in immunoreactivity of AR between the breeding and nonbreeding seasons. However, the immunoreactivities of ERa and ERb were both significantly reduced in the nonbreeding season compared to the breeding season. The positive stains of FSHR and LHR were found in the granulosa cells and theca cells of the ovaries of the breeding and nonbreeding seasons. In addition, the Western blotting results of FSHR and LHR showed a significant reduction in the nonbreeding season compared with the breeding season. These findings suggested that androgen might be predominantly converted into estrogen in order to regulate the follicular development via binding of estrogen receptors during the breeding season, whereas androgen might predominantly directly bind androgen receptor to regulate the follicular development during the nonbreeding season in the ovaries of wild female ground squirrels.
Figures


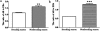
Similar articles
-
Seasonal expression of androgen receptor, aromatase, and estrogen receptor alpha and beta in the testis of the wild ground squirrel (Citellus dauricus Brandt).Eur J Histochem. 2015 Feb 17;59(1):2456. doi: 10.4081/ejh.2015.2456. Eur J Histochem. 2015. PMID: 25820559 Free PMC article.
-
Seasonal expressions of androgen receptor, estrogen receptors and cytochrome P450 aromatase in the uteri of the wild Daurian ground squirrels (Spermophilus dauricus).Eur J Histochem. 2018 Feb 7;62(1):2889. doi: 10.4081/ejh.2018.2889. Eur J Histochem. 2018. PMID: 29569876 Free PMC article.
-
Seasonal changes of androgen receptor, estrogen receptors and aromatase expression in the hippocampus of the wild male ground squirrels (Citellus dauricus Brandt).Gen Comp Endocrinol. 2017 Aug 1;249:93-100. doi: 10.1016/j.ygcen.2017.05.009. Epub 2017 May 11. Gen Comp Endocrinol. 2017. PMID: 28502742
-
Seasonal changes in spermatogenesis and immunolocalization of cytochrome P450 17alpha-hydroxylase/c17-20 lyase and cytochrome P450 aromatase in the wild male ground squirrel (Citellus dauricus Brandt).J Reprod Dev. 2010 Jun;56(3):297-302. doi: 10.1262/jrd.09-078t. Epub 2010 Mar 3. J Reprod Dev. 2010. PMID: 20197644
-
Differential regulation of aromatase and androgen receptor in granulosa cells.J Steroid Biochem Mol Biol. 1997 Apr;61(3-6):233-9. J Steroid Biochem Mol Biol. 1997. PMID: 9365195 Review.
Cited by
-
Caloric restriction increases ratio of estrogen to androgen receptors expression in murine ovaries--potential therapeutic implications.J Ovarian Res. 2015 Aug 13;8:57. doi: 10.1186/s13048-015-0185-8. J Ovarian Res. 2015. PMID: 26264910 Free PMC article.
-
Seasonal changes in expression of nerve growth factor and its receptors TrkA and p75 in the ovary of wild ground squirrel (Citellus dauricus Brandt).J Ovarian Res. 2014 Jan 9;7:3. doi: 10.1186/1757-2215-7-3. J Ovarian Res. 2014. PMID: 24405743 Free PMC article.
-
Seasonal expression of androgen receptor, aromatase, and estrogen receptor alpha and beta in the testis of the wild ground squirrel (Citellus dauricus Brandt).Eur J Histochem. 2015 Feb 17;59(1):2456. doi: 10.4081/ejh.2015.2456. Eur J Histochem. 2015. PMID: 25820559 Free PMC article.
-
Seasonal changes of androgen receptor, estrogen receptors and aromatase expression in the medial preoptic area of the wild male ground squirrels (Citellus dauricus Brandt).Eur J Histochem. 2016 May 2;60(2):2621. doi: 10.4081/ejh.2016.2621. Eur J Histochem. 2016. PMID: 27349316 Free PMC article.
-
Seasonal patterns of prolactin, prolactin receptor, and STAT5 expression in the ovaries of wild ground squirrels (<em>Citellus dauricus</em> Brandt).Eur J Histochem. 2023 Oct 2;67(4):3825. doi: 10.4081/ejh.2023.3825. Eur J Histochem. 2023. PMID: 37781865 Free PMC article.
References
-
- Couse JF, Yates MM, Walker VR, Korach KS. Characterization of the hypothalamic-pituitary-gonadal axis in estrogen receptor (ER) Null mice reveals hypergonadism and endocrine sex reversal in females lacking ERalpha but not ERbeta. Mol Endocrinol. 2003;17:1039–1053. doi: 10.1210/me.2002-0398. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

