Lymphocyte depletion in experimental hemorrhagic shock in Swine
- PMID: 23009382
- PMCID: PMC3551698
- DOI: 10.1186/1476-9255-9-34
Lymphocyte depletion in experimental hemorrhagic shock in Swine
Abstract
Background: Hemorrhagic shock results in systemic activation of the immune system and leads to ischemia-reperfusion injury. Lymphocytes have been identified as critical mediators of the early innate immune response to ischemia-reperfusion injury, and immunomodulation of lymphocytes may prevent secondary immunologic injury in surgical and trauma patients.
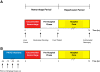
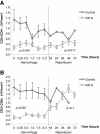
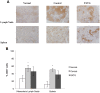
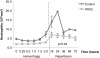
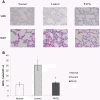
Methods: Yorkshire swine were anesthetized and underwent a grade III liver injury with uncontrolled hemorrhage to induce hemorrhagic shock. Experimental groups were treated with a lymphocyte depletional agent, porcine polyclonal anti-thymocyte globulin (PATG) (n = 8) and compared to a vehicle control group (n = 9). Animals were observed over a 3 day survival period. Circulating lymphocytes were examined with FACS analysis for CD3/CD4/CD8, and central lymphocytes with mesenteric lymph node and spleen staining for CD3. Circulating and lung tissue16 infiltrating neutrophils were measured. Circulating CD3 lymphocytes in the blood and in central lymphoid organs (spleen/lymph node) were stained and evaluated using FACS analysis. Immune-related gene expression from liver tissue was quantified using RT-PCR.
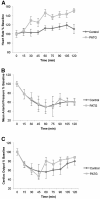
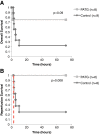
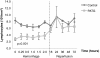
Results: The overall survival was 22% (2/9) in the control and 75% (6/8) in the PATG groups, p = 0.09; during the reperfusion period (following hemorrhage) survival was 25% (2/8) in the control and 100% (6/6) in the PATG groups, p = 0.008. Mean blood loss and hemodynamic profiles were not significantly different between the experimental and control groups. Circulating CD3+CD4+ lymphocytes were significantly depleted in the PATG group compared to control. Lymphocyte depletion in the setting of hemorrhagic shock also significantly decreased circulating and lung tissue infiltrating neutrophils, and decreased expression of liver ischemia gene expression.
Conclusions: Lymphocyte manipulation with a depletional (PATG) strategy improves reperfusion survival in experimental hemorrhagic shock using a porcine liver injury model. This proof of principle study paves the way for further development of immunomodulation approaches to ameliorate secondary immune injury following hemorrhagic shock.
Figures
Similar articles
-
Lymphocyte modulation with FTY720 improves hemorrhagic shock survival in swine.PLoS One. 2012;7(4):e34224. doi: 10.1371/journal.pone.0034224. Epub 2012 Apr 30. PLoS One. 2012. PMID: 22558085 Free PMC article.
-
Lymphodepletive effects of rabbit anti-pig thymocyte globulin in neonatal swines.Transpl Immunol. 2016 Nov;39:74-83. doi: 10.1016/j.trim.2016.08.005. Epub 2016 Aug 22. Transpl Immunol. 2016. PMID: 27561237
-
Immune potential of lymph node-derived lymphocytes in uncontrolled hemorrhagic shock.Shock. 2001 Feb;15(2):118-23. doi: 10.1097/00024382-200115020-00007. Shock. 2001. PMID: 11220639
-
[Induction mechanism of shock: applying the etiology in judgment of the cause of death in forensic practice].Nihon Hoigaku Zasshi. 2004 Sep;58(2):130-40. Nihon Hoigaku Zasshi. 2004. PMID: 15526767 Review. Japanese.
-
[Deep lung--cellular reaction to HIV].Rev Port Pneumol. 2007 Mar-Apr;13(2):175-212. Rev Port Pneumol. 2007. PMID: 17492233 Review. Portuguese.
Cited by
-
Immunopathological Alterations after Blast Injury and Hemorrhage in a Swine Model of Prolonged Damage Control Resuscitation.Int J Mol Sci. 2023 Apr 19;24(8):7494. doi: 10.3390/ijms24087494. Int J Mol Sci. 2023. PMID: 37108656 Free PMC article.
-
A comprehensive overview of fixed-volume hemorrhage effects in New Zealand White rabbit models.Open Vet J. 2025 Mar;15(3):1253-1263. doi: 10.5455/OVJ.2025.v15.i3.17. Epub 2025 Mar 31. Open Vet J. 2025. PMID: 40276174 Free PMC article.
-
Navigating Hemorrhagic Shock: Biomarkers, Therapies, and Challenges in Clinical Care.Biomedicines. 2024 Dec 17;12(12):2864. doi: 10.3390/biomedicines12122864. Biomedicines. 2024. PMID: 39767770 Free PMC article. Review.
References
-
- Moore FA, Moore EE. Postinjury multiple organ failure. Trauma. 5. New York: McGraw-Hill; 2004.
-
- Rhee P, Morris J, Durham R, Hauser C, Cipolle M, Wilson R, Luchette F, McSwain N, Miller R. Recombinant humanized monoclonal antibody against CD18 (rhuMAb CD18) in traumatic hemorrhagic shock: results of a phase II clinical trial. Traumatic Shock Group. J Trauma. 2000;49:611–619. doi: 10.1097/00005373-200010000-00007. discussion 619–620. - DOI - PubMed
-
- Yonekawa K, Harlan JM. Targeting leukocyte integrins in human diseases. J Leukoc Biol. 2005;77:129–140. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

