Pharmacoeconomics and its implication on priority-setting for essential medicines in Tanzania: a systematic review
- PMID: 23016739
- PMCID: PMC3472274
- DOI: 10.1186/1472-6947-12-110
Pharmacoeconomics and its implication on priority-setting for essential medicines in Tanzania: a systematic review
Abstract
Background: Due to escalating treatment costs, pharmacoeconomic analysis has been assigned a key role in the quest for increased efficiency in resource allocation for drug therapies in high-income countries. The extent to which pharmacoeconomic analysis is employed in the same role in low-income countries is less well established. This systematic review identifies and briefly describes pharmacoeconomic studies which have been conducted in Tanzania and further assesses their influence in the selection of essential medicines.
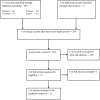
Methods: Pubmed, Embase, Cinahl and Cochrane databases were searched using "economic evaluation", "cost-effectiveness analysis", "cost-benefit analysis" AND "Tanzania" as search terms. We also scanned reference lists and searched in Google to identify other relevant articles. Only articles reporting full economic evaluations about drug therapies and vaccines conducted in Tanzania were included. The national essential medicine list and other relevant policy documents related to the identified articles were screened for information regarding the use of economic evaluation as a criterion for medicine selection.
Results: Twelve pharmacoeconomic studies which met our inclusion criteria were identified. Seven studies were on HIV/AIDS, malaria and diarrhoea, the three highest ranked diseases on the disease burden in Tanzania. Six studies were on preventive and treatment interventions targeting pregnant women and children under the age of five years. The national essential medicine list and the other identified policy documents do not state the use of economic evaluation as one of the criteria which has influenced the listing of the drugs.
Conclusion: Country specific pharmacoeconomic analyses are too scarce and inconsistently used to have had a significant influence on the selection of essential medicines in Tanzania. More studies are required to fill the existing gap and to explore whether decision-makers have the ability to interpret and utilise pharmacoeconomic evidence. Relevant health authorities in Tanzania should also consider how to apply pharmacoeconomic analyses more consistently in the future priority-setting decisions for selection of essential medicines.
Figures
References
-
- Drummond MF, Sculpher MJ, Torrance GW, O’Brien BJ, Stoddart GL. Methods for the Economic Evaluation of Health Care Programmes. 3. New York: Oxford University Press; 2005.
-
- National Institute for Health and Clinical Excellence. Guide to the methods of technology appraisal. London; 2008. - PubMed
-
- CADTH. Guidelines for the economic evaluation of health technologies. 3. Ottawa, Canada: Canadian Agency for Drugs and Technologies in Health; 2006.
-
- Sanchez LA. Pharmacoeconomics and formulary decision-making. PharmacoEconomics. 1996;9(suppl 1):16–25. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous