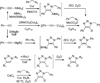
Synthesis of deuterated 1,2,3-triazoles
- PMID: 23016757
- PMCID: PMC3543115
- DOI: 10.1021/jo301146j
Synthesis of deuterated 1,2,3-triazoles
Abstract
The copper-catalyzed azide-alkyne cycloaddition (CuAAC) is a highly effective method for the selective incorporation of deuterium atom into the C-5 position of the 1,2,3-triazole structure. Reactions of alkynes and azides can be conveniently carried out in a biphasic medium of CH(2)Cl(2)/D(2)O, using the CuSO(4)/Na ascorbate system. The mildness of the method renders it applicable to substrates of relatively high complexity, such as nucleosides. Good yields and high levels of deuterium incorporation were observed. A reaction conducted in equimolar H(2)O and D(2)O showed 2.7 times greater incorporation of hydrogen atom as compared to deuterium. This is consistent with the H(+) and D(+) ion concentrations in H(2)O and D(2)O, respectively. With appropriately deuterated precursors, partially to fully deuterated triazoles were assembled where the final deuterium atom was incorporated in the triazole-forming step.
Figures
References
-
- Huisgen R, Szeimies G, Möbius L. Chem. Ber. 1967;100:2492–2507.
- Huisgen R, Knorr R, Möbius L, Szeimies G. Chem. Ber. 1965;98:4041–4021.
- Huisgen R. Angew. Chem. Int. Ed. 1963;2:633–645.
- Huisgen R. Angew. Chem. Int. Ed. 1963;2:565–598.
-
- Tornøe CW, Christensen C, Meldal M. J. Org. Chem. 2002;67:3057–3064. - PubMed
-
- Rostovtsev VV, Green LG, Fokin VV, Sharpless KB. Angew. Chem. Int. Ed. 2002;41:2596–2599. - PubMed
-
-
For reviews see:
- Kolb HC, Finn MG, Sharpless KB. Angew. Chem. Int. Ed. 2001;40:2004–2021. - PubMed
- Gil MV, Arévalo MJ, López O. Synthesis. 2007:1589–1620.
-
-
- Ahlquist M, Fokin VV. Organometallics. 2007;26:4389–4391.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous