Rejuvenating sirtuins: the rise of a new family of cancer drug targets
- PMID: 23016857
- PMCID: PMC3549556
- DOI: 10.2174/138161213804581954
Rejuvenating sirtuins: the rise of a new family of cancer drug targets
Abstract
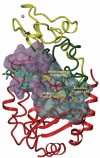
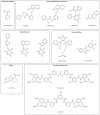
Sirtuins are a family of NAD+-dependent enzymes that was proposed to control organismal life span about a decade ago. While such role of sirtuins is now debated, mounting evidence involves these enzymes in numerous physiological processes and disease conditions, including metabolism, nutritional behavior, circadian rhythm, but also inflammation and cancer. SIRT1, SIRT2, SIRT3, SIRT6, and SIRT7 have all been linked to carcinogenesis either as tumor suppressor or as cancer promoting proteins. Here, we review the biological rationale for the search of sirtuin inhibitors and activators for treating cancer and the experimental approaches to their identification.
Figures

References
-
- Frye RA. Characterization of five human cDNAs with homology to the yeast SIR2 gene: Sir2-like proteins (sirtuins) metabolize NAD and may have protein ADP-ribosyltransferase activity. Biochem Biophys Res Commun. 1999;260:273–9. - PubMed
-
- Tanny JC, Dowd GJ, Huang J, Hilz H, Moazed D. An enzymatic activity in the yeast Sir2 protein that is essential for gene silencing. Cell. 1999; 99:735–45. - PubMed
-
- Imai S, Armstrong CM, Kaeberlein M, Guarente L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature. 2000; 403:795–800. - PubMed
-
- Dani N, Barbosa AJM, Del Rio A, Di Girolamo M. ADPribosylated proteins as old and new drug targets for anticancer therapy: the example of ARF6. Curr Pharm Des. 2013;19(4):624–33. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

