Clinically employed opioid analgesics produce antinociception via μ-δ opioid receptor heteromers in Rhesus monkeys
- PMID: 23019498
- PMCID: PMC3447399
- DOI: 10.1021/cn300049m
Clinically employed opioid analgesics produce antinociception via μ-δ opioid receptor heteromers in Rhesus monkeys
Abstract
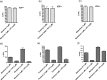
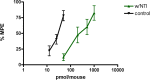
Morphine and related drugs are widely employed as analgesics despite the side effects associated with their use. Although morphine is thought to mediate analgesia through mu opioid receptors, delta opioid receptors have been implicated in mediating some side effects such as tolerance and dependence. Here we present evidence in rhesus monkeys that morphine, fentanyl, and possibly methadone selectively activate mu-delta heteromers to produce antinociception that is potently antagonized by the delta opioid receptor antagonist, naltrindole (NTI). Studies with HEK293 cells expressing mu-delta heteromeric opioid receptors exhibit a similar antagonism profile of receptor activation in the presence of NTI. In mice, morphine was potently inhibited by naltrindole when administered intrathecally, but not intracerebroventricularly, suggesting the possible involvement of mu-delta heteromers in the spinal cord of rodents. Taken together, these results strongly suggest that, in primates, mu-delta heteromers are allosterically coupled and mediate the antinociceptive effects of three clinically employed opioid analgesics that have been traditionally viewed as mu-selective. Given the known involvement of delta receptors in morphine tolerance and dependence, our results implicate mu-delta heteromers in mediating both antinociception and these side effects in primates. These results open the door for further investigation in humans.
Figures
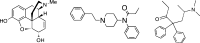


Similar articles
-
Opioid interactions in rhesus monkeys: effects of delta + mu and delta + kappa agonists on schedule-controlled responding and thermal nociception.J Pharmacol Exp Ther. 2003 Dec;307(3):1054-64. doi: 10.1124/jpet.103.056515. Epub 2003 Oct 13. J Pharmacol Exp Ther. 2003. PMID: 14557380
-
The effect of chronic treatment with naltrindole, a selective delta-opioid antagonist, on mu-opioid receptor-mediated antinociception in diabetic mice.Psychopharmacology (Berl). 1993;113(2):167-71. doi: 10.1007/BF02245693. Psychopharmacology (Berl). 1993. PMID: 7855177
-
Delta opioid receptor enhancement of mu opioid receptor-induced antinociception in spinal cord.J Pharmacol Exp Ther. 1998 Jun;285(3):1181-6. J Pharmacol Exp Ther. 1998. PMID: 9618421
-
Evidence and Function Relevance of Native DOR-MOR Heteromers.Handb Exp Pharmacol. 2018;247:115-127. doi: 10.1007/164_2018_112. Handb Exp Pharmacol. 2018. PMID: 29633181 Review.
-
Opioid receptor heteromers in analgesia.Expert Rev Mol Med. 2012 Apr 10;14:e9. doi: 10.1017/erm.2012.5. Expert Rev Mol Med. 2012. PMID: 22490239 Free PMC article. Review.
Cited by
-
Mu Opioid Receptor Heterodimers Emerge as Novel Therapeutic Targets: Recent Progress and Future Perspective.Front Pharmacol. 2020 Jul 15;11:1078. doi: 10.3389/fphar.2020.01078. eCollection 2020. Front Pharmacol. 2020. PMID: 32760281 Free PMC article. Review.
-
Rubiscolin-6 activates opioid receptors to enhance glucose uptake in skeletal muscle.J Food Drug Anal. 2019 Jan;27(1):266-274. doi: 10.1016/j.jfda.2018.06.012. Epub 2018 Aug 14. J Food Drug Anal. 2019. PMID: 30648580 Free PMC article.
-
Putative kappa opioid heteromers as targets for developing analgesics free of adverse effects.J Med Chem. 2014 Aug 14;57(15):6383-92. doi: 10.1021/jm500159d. Epub 2014 Jul 18. J Med Chem. 2014. PMID: 24978316 Free PMC article.
-
Heteroatom analogues of hydrocodone: synthesis and biological activity.J Org Chem. 2013 Apr 5;78(7):2914-25. doi: 10.1021/jo3026753. Epub 2013 Mar 6. J Org Chem. 2013. PMID: 23397939 Free PMC article.
-
Heteromers of μ-δ opioid receptors: new pharmacology and novel therapeutic possibilities.Br J Pharmacol. 2015 Jan;172(2):375-87. doi: 10.1111/bph.12663. Epub 2014 Jul 1. Br J Pharmacol. 2015. PMID: 24571499 Free PMC article. Review.
References
-
- Dubois M. Y.; Gallagher R. M.; Lippe P. M. (2009) Pain medicine position paper. Pain Med. 10(6), 972–1000. - PubMed
-
- Gutstein H. B., Akil H. (2006) Opioid analgesics, in Goodman & Gilman's The Pharmacological Basis of Therapeutics (Brunton L. L., Lazo J. S., Parker K. L., Eds.) 11th ed., pp 547–90, McGraw Hill, New York.
-
- Bowen C. A.; Fischer B. D.; Mello N. K.; Negus S. S. (2002) Antagonism of the antinociceptive and discriminative stimulus effects of heroin and morphine by 3-methoxynaltrexone and naltrexone in rhesus monkeys. J. Pharmacol. Exp. Ther. 302(1), 264–73. - PubMed
-
- Emmerson P. J.; Liu M. R.; Woods J. H.; Medzihradsky F. (1994) Binding affinity and selectivity of opioids at mu, delta and kappa receptors in monkey brain membranes. J. Pharmacol. Exp. Ther. 271(3), 1630–7. - PubMed
-
- Emmerson P. J.; Clark M. J.; Mansour A.; Akil H.; Woods J. H.; Medzihradsky F. (1996) Characterization of opioid agonist efficacy in a C6 glioma cell line expressing the mu opioid receptor. J. Pharmacol. Exp. Ther. 278(3), 1121–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

