Protein tyrosine kinase 6 protects cells from anoikis by directly phosphorylating focal adhesion kinase and activating AKT
- PMID: 23027128
- PMCID: PMC3940264
- DOI: 10.1038/onc.2012.427
Protein tyrosine kinase 6 protects cells from anoikis by directly phosphorylating focal adhesion kinase and activating AKT
Abstract
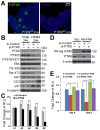
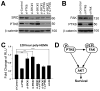
Protein tyrosine kinase 6 (PTK6) is a non-receptor tyrosine kinase expressed in epithelial cancers. Disruption of Ptk6 decreases azoxymethane-induced colon tumorigenesis in mice by preventing signal transducer and activator of transcription 3 activation. Relocalization of PTK6 in prostate cancers contributes to increased growth. Although not expressed in normal breast or ovary, PTK6 promotes anchorage-independent survival of breast and ovarian tumor cells. We identified several potential PTK6 substrates in the human SW620 colon cancer cell line using mass spectrometry, including FAK (focal adhesion kinase). We show that FAK is a direct substrate of PTK6 in vitro and in vivo. Expression of membrane-targeted active PTK6 (Palm-PTK6-YF) induces constitutive activation of FAK and cell morphology changes, which are independent of SRC family kinases in Src-/-, Yes-/-, Fyn-/- (SYF) mouse embryonic fibroblasts (MEFs). Palm-PTK6-YF expressing SYF cells are transformed and overcome contact inhibition, form colonies in transformation assays, proliferate in suspension and form tumors in a xenograft model. Expression of FAK and Palm-PTK6-YF in Fak-/- MEFs synergistically activates AKT and protects cells against anoikis. However, expression of Palm-PTK6-YF in Akt1/2-/- MEFs fails to protect cells from anoikis, indicating AKT is critical in PTK6 and FAK-mediated survival signaling. In a conditional Pten knockout murine prostate cancer model, we identify prostate epithelial cells with enhanced activation of endogenous PTK6 and FAK at the plasma membrane. Knockdown of PTK6 in the PC3 human prostate cancer cell line disrupts FAK and AKT activation and promotes anoikis, which can be rescued by exogenous expression of FAK. Our data reveal important roles for a PTK6-FAK-AKT signaling axis in promoting anchorage-independent cell survival.
Conflict of interest statement
Conflict of interest.
The authors declare no conflict of interest.
Figures




References
-
- Mitchell PJ, Barker KT, Martindale JE, Kamalati T, Lowe PN, Page MJ, et al. Cloning and characterisation of cDNAs encoding a novel non-receptor tyrosine kinase, brk, expressed in human breast tumours. Oncogene. 1994;9:2383–90. - PubMed
-
- Siyanova EY, Serfas MS, Mazo IA, Tyner AL. Tyrosine kinase gene expression in the mouse small intestine. Oncogene. 1994;9(7):2053–7. - PubMed
-
- Derry JJ, Prins GS, Ray V, Tyner AL. Altered localization and activity of the intracellular tyrosine kinase BRK/Sik in prostate tumor cells. Oncogene. 2003 Jul 3;22(27):4212–20. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

