Golgi phosphoprotein 3 determines cell binding properties under dynamic flow by controlling Golgi localization of core 2 N-acetylglucosaminyltransferase 1
- PMID: 23027862
- PMCID: PMC3501027
- DOI: 10.1074/jbc.M112.346528
Golgi phosphoprotein 3 determines cell binding properties under dynamic flow by controlling Golgi localization of core 2 N-acetylglucosaminyltransferase 1
Abstract
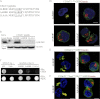
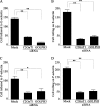
Core 2 N-acetylglucosaminyltransferase 1 (C2GnT1) is a key enzyme participating in the synthesis of core 2-associated sialyl Lewis x (C2-O-sLe(x)), a ligand involved in selectin-mediated leukocyte trafficking and cancer metastasis. To accomplish that, C2GnT1 needs to be localized to the Golgi and this step requires interaction of its cytoplasmic tail (CT) with a protein that has not been identified. Employing C2GnT1 CT as the bait to perform a yeast two-hybrid screen, we have identified Golgi phosphoprotein 3 (GOLPH3) as a principal candidate protein that interacts with C2GnT1 and demonstrated that C2GnT1 binds to GOLPH3 via the LLRRR(9) sequence in the CT. Confocal fluorescence microscopic analysis shows substantial Golgi co-localization of C2GnT1 and GOLPH3. Upon GOLPH3 knockdown, C2GnT1 is found mainly in the endoplasmic reticulum and decorated with complex-type N-glycans, indicating that the enzyme has been transported to the Golgi but is not retained. Also, we have found that a recombinant protein consisting of C2GnT1 CT(1-16)-Leu(17-32)-Gly(33-42)-GFP is localized to the Golgi although the same construct with mutated CT (AAAAA(9)) is not. The data demonstrate that the C2GnT1 CT is necessary and sufficient for Golgi localization of C2GnT1. Furthermore, GOLPH3 knockdown results in reduced synthesis of C2-O-sLe(x) associated with P-selectin glycoprotein ligand-1, reduced cell tethering to and rolling on immobilized P- or E-selectin, and compromised E-selectin-induced activation of spleen tyrosine kinase and cell adhesion to intercellular adhesion molecule-1 under dynamic flow. Our results reveal that GOLPH3 can regulate cell-cell interaction by controlling Golgi retention of C2GnT1.
Figures







References
-
- van den Berg T. K., Döpp E. A., Dijkstra C. D. (2001) Rat macrophages. Membrane glycoproteins in differentiation and function. Immunol. Rev. 184, 45–57 - PubMed
-
- Feizi T. (1980) Structural and biological aspects of blood group I and i antigens on glycolipids and glycoproteins. Rev. Fr. Transfus. Immunohematol. 23, 563–577 - PubMed
-
- Ochs H. D., Wedgwood R. J., Heller S. R., Beatty P. G. (1986) Complement, membrane glycoproteins, and complement receptors. Their role in regulation of the immune response. Clin. Immunol. Immunopathol. 40, 94–104 - PubMed
-
- Chen S., Fukuda M. (2006) Cell type-specific roles of carbohydrates in tumor metastasis. Methods Enzymol. 416, 371–380 - PubMed
-
- Varki A. C. R., Esko J., Freeze H., Stanley P., Bertozzi C., Hart G., Etzler M. (2009) Essentials of Glycobiology, 2nd Ed., Cold Spring Harbor Laboratory, Cold Spring Harbor, NY - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

