Cell autonomous lipin 1 function is essential for development and maintenance of white and brown adipose tissue
- PMID: 23028044
- PMCID: PMC3497595
- DOI: 10.1128/MCB.00512-12
Cell autonomous lipin 1 function is essential for development and maintenance of white and brown adipose tissue
Abstract
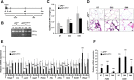
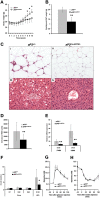
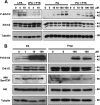
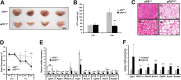
Through analysis of mice with spatially and temporally restricted inactivation of Lpin1, we characterized its cell autonomous function in both white (WAT) and brown (BAT) adipocyte development and maintenance. We observed that the lipin 1 inactivation in adipocytes of aP2(Cre/+)/Lp(fEx2)(-)(3/fEx2)(-)(3) mice resulted in lipodystrophy and the presence of adipocytes with multilocular lipid droplets. We further showed that time-specific loss of lipin 1 in mature adipocytes in aP2(Cre-ERT2/+)/Lp(fEx2)(-)(3/fEx2)(-)(3) mice led to their replacement by newly formed Lpin1-positive adipocytes, thus establishing a role for lipin 1 in mature adipocyte maintenance. Importantly, we observed that the presence of newly formed Lpin1-positive adipocytes in aP2(Cre-ERT2/+)/Lp(fEx2)(-)(3/fEx2)(-)(3) mice protected these animals against WAT inflammation and hepatic steatosis induced by a high-fat diet. Loss of lipin 1 also affected BAT development and function, as revealed by histological changes, defects in the expression of peroxisome proliferator-activated receptor alpha (PPARα), PGC-1α, and UCP1, and functionally by altered cold sensitivity. Finally, our data indicate that phosphatidic acid, which accumulates in WAT of animals lacking lipin 1 function, specifically inhibits differentiation of preadipocytes. Together, these observations firmly demonstrate a cell autonomous role of lipin 1 in WAT and BAT biology and indicate its potential as a therapeutical target for the treatment of obesity.
Figures





References
-
- Abumrad NA, el-Maghrabi MR, Amri EZ, Lopez E, Grimaldi PA. 1993. Cloning of a rat adipocyte membrane protein implicated in binding or transport of long-chain fatty acids that is induced during preadipocyte differentiation. Homology with human CD36. J. Biol. Chem. 268: 17665– 17668 - PubMed
-
- Andresen BT, Rizzo MA, Shome K, Romero G. 2002. The role of phosphatidic acid in the regulation of the Ras/MEK/Erk signaling cascade. FEBS Lett. 531: 65– 68 - PubMed
-
- Bjorntorp P, et al. 1978. Isolation and characterization of cells from rat adipose tissue developing into adipocytes. J. Lipid Res. 19: 316– 324 - PubMed
-
- Bost F, Aouadi M, Caron L, Binetruy B. 2005. The role of MAPKs in adipocyte differentiation and obesity. Biochimie 87: 51– 56 - PubMed
-
- Cancello R, et al. 2006. Increased infiltration of macrophages in omental adipose tissue is associated with marked hepatic lesions in morbid human obesity. Diabetes 55: 1554– 1561 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
