Cardiac regeneration from activated epicardium
- PMID: 23028582
- PMCID: PMC3447884
- DOI: 10.1371/journal.pone.0044692
Cardiac regeneration from activated epicardium
Abstract
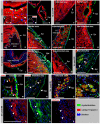
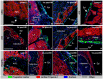
In contrast to lower vertebrates, the mammalian heart has a very limited regenerative capacity. Cardiomyocytes, lost after ischemia, are replaced by fibroblasts. Although the human heart is able to form new cardiomyocytes throughout its lifespan, the efficiency of this phenomenon is not enough to substitute sufficient myocardial mass after an infarction. In contrast, zebrafish hearts regenerate through epicardial activation and initiation of myocardial proliferation. With this study we obtain insights into the activation and cellular contribution of the mammalian epicardium in response to ischemia. In a mouse myocardial infarction model we analyzed the spatio-temporal changes in expression of embryonic epicardial, EMT, and stem cell markers and the contribution of cells of the Wt1-lineage to the infarcted area. Though the integrity of the epicardial layer overlaying the infarct is lost immediately after the induction of the ischemia, it was found to be regenerated at three days post infarction. In this regenerated epicardium, the embryonic gene program is transiently re-expressed as well as proliferation. Concomitant with this activation, Wt1-lineage positive subepicardial mesenchyme is formed until two weeks post-infarction. These mesenchymal cells replace the cardiomyocytes lost due to the ischemia and contribute to the fibroblast population, myofibroblasts and coronary endothelium in the infarct, and later also to the cardiomyocyte population. We show that in mice, as in lower vertebrates, an endogenous, epicardium-dependent regenerative response to injury is induced. Although this regenerative response leads to the formation of new cardiomyocytes, their number is insufficient in mice but sufficient in lower vertebrates to replace lost cardiomyocytes. These molecular and cellular analyses provide basic knowledge essential for investigations on the regeneration of the mammalian heart aiming at epicardium-derived cells.
Conflict of interest statement
Figures





References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

