Impaired myogenesis in estrogen-related receptor γ (ERRγ)-deficient skeletal myocytes due to oxidative stress
- PMID: 23038752
- PMCID: PMC3528312
- DOI: 10.1096/fj.12-212290
Impaired myogenesis in estrogen-related receptor γ (ERRγ)-deficient skeletal myocytes due to oxidative stress
Abstract
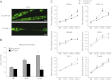
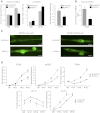
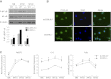
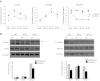
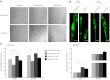
Specialized contractile function and increased mitochondrial number and oxidative capacity are hallmark features of myocyte differentiation. The estrogen-related receptors (ERRs) can regulate mitochondrial biogenesis or mitochondrial enzyme expression in skeletal muscle, suggesting that ERRs may have a role in promoting myogenesis. Therefore, we characterized myogenic programs in primary myocytes isolated from wild-type (M-ERRγWT) and muscle-specific ERRγ(-/-) (M-ERRγ(-/-)) mice. Myotube maturation and number were decreased throughout differentiation in M-ERRγ(-/-) primary myocytes, resulting in myotubes with reduced mitochondrial content and sarcomere assembly. Compared with M-ERRγWT myocytes at the same differentiation stage, the glucose oxidation rate was reduced by 30% in M-ERRγ(-/-) myotubes, while medium-chain fatty acid oxidation was increased by 34% in M-ERRγ(-/-) myoblasts and 36% in M-ERRγ(-/-) myotubes. Concomitant with increased reliance on mitochondrial β-oxidation, H(2)O(2) production was significantly increased by 40% in M-ERRγ(-/-) myoblasts and 70% in M-ERRγ(-/-) myotubes compared to M-ERRγWT myocytes. ROS activation of FoxO and NF-κB and their downstream targets, atrogin-1 and MuRF1, was observed in M-ERRγ(-/-) myocytes. The antioxidant N-acetyl cysteine rescued myotube formation and atrophy gene induction in M-ERRγ(-/-) myocytes. These results suggest that loss of ERRγ causes metabolic defects and oxidative stress that impair myotube formation through activation of skeletal muscle atrophy pathways.
Figures



References
-
- Huss J. M., Kopp R. P., Kelly D. P. (2002) Peroxisome proliferator-activated receptor coactivator-1alpha (PGC-1alpha) coactivates the cardiac-enriched nuclear receptors estrogen-related receptor-alpha and -gamma. Identification of novel leucine-rich interaction motif within PGC-1alpha. J. Biol. Chem. 277, 40265–40274 - PubMed
-
- Kamei Y., Ohizumi H., Fujitani Y., Nemoto T., Tanaka T., Takahashi N., Kawada T., Miyoshi M., Ezaki O., Kakizuka A. (2003) PPARgamma coactivator 1β/ERR ligand 1 is an ERR protein ligand, whose expression induces a high-energy expenditure and antagonizes obesity. Proc. Natl. Acad. Sci. U. S. A. 100, 12378–12383 - PMC - PubMed
-
- Laganiere J., Tremblay G. B., Dufour C. R., Giroux S., Rousseau F., Giguere V. (2004) A polymorphic autoregulatory hormone response element in the human estrogen-related receptor alpha (ERRα) promoter dictates peroxisome proliferator-activated receptor gamma coactivator-1alpha control of ERRalpha expression. J. Biol. Chem. 279, 18504–18510 - PubMed
-
- Schreiber S. N., Emter R., Hock M. B., Knutti D., Cardenas J., Podvinec M., Oakeley E. J., Kralli A. (2004) The estrogen-related receptor alpha (ERRalpha) functions in PPARgamma coactivator 1alpha (PGC-1α)-induced mitochondrial biogenesis. Proc. Natl. Acad. Sci. U. S. A. 101, 6472–6477 - PMC - PubMed
-
- Dufour C. R., Wilson B. J., Huss J. M., Kelly D. P., Alaynick W. A., Downes M., Evans R. M., Blanchette M., Giguere V. (2007) Genome-wide orchestration of cardiac functions by the orphan nuclear receptors ERRalpha and gamma. Cell Metab. 5, 345–356 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

