Non-monotonic dose effects of in utero exposure to di(2-ethylhexyl) phthalate (DEHP) on testicular and serum testosterone and anogenital distance in male mouse fetuses
- PMID: 23041310
- PMCID: PMC3543148
- DOI: 10.1016/j.reprotox.2012.09.006
Non-monotonic dose effects of in utero exposure to di(2-ethylhexyl) phthalate (DEHP) on testicular and serum testosterone and anogenital distance in male mouse fetuses
Abstract
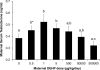
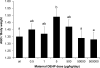
Di(2-ethylhexyl) phthalate (DEHP) is a ubiquitous environmental contaminant. Epidemiological studies suggest that DEHP decreases masculinization of male fetuses. Numerous rat studies report DEHP reduces fetal testosterone production at doses greatly exceeding human exposure. We fed pregnant CD-1 mice 0.5-500,000 μg/kg/day DEHP from gestation day (GD) 9-18 and examined mothers and male fetuses on GD 18. We assessed non-monotonic dose-response by adding a quadratic term to a simple linear regression model. Except at the 500,000 μg/kg/day dose, DEHP stimulated an increase in maternal and fetal serum testosterone and increased anogenital distance (AGD). Non-monotonic dose-response curves were noted for AGD and maternal, and testis testosterone (P values 0.013-0.021). Because data from our highest dose (500,000 μg/kg/day) did not differ significantly from controls, this dose could have been incorrectly assumed to be the NOAEL had we only tested very high doses, as is typical in studies for regulatory agencies.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures

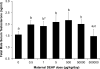
Similar articles
-
Gestational and pubertal exposure to low dose of di-(2-ethylhexyl) phthalate impairs sperm quality in adult mice.Reprod Toxicol. 2020 Sep;96:175-184. doi: 10.1016/j.reprotox.2020.06.014. Epub 2020 Jun 30. Reprod Toxicol. 2020. PMID: 32619501
-
Disruption of reproductive development in male rat offspring following gestational and lactational exposure to di-(2-ethylhexyl) phthalate and genistein.Biol Res. 2013;46(2):139-46. doi: 10.4067/S0716-97602013000200004. Biol Res. 2013. PMID: 23959011
-
Effects of In Utero and Lactational Exposure to New Generation Green Plasticizers on Adult Male Rats: A Comparative Study With Di(2-Ethylhexyl) Phthalate.Toxicol Sci. 2018 Jul 1;164(1):129-141. doi: 10.1093/toxsci/kfy072. Toxicol Sci. 2018. PMID: 29945229 Free PMC article.
-
Systematic reviews and meta-analyses of human and animal evidence of prenatal diethylhexyl phthalate exposure and changes in male anogenital distance.J Toxicol Environ Health B Crit Rev. 2018;21(4):207-226. doi: 10.1080/10937404.2018.1505354. Epub 2018 Sep 10. J Toxicol Environ Health B Crit Rev. 2018. PMID: 30199328 Free PMC article.
-
Fetal origin of endocrine dysfunction in the adult: the phthalate model.J Steroid Biochem Mol Biol. 2013 Sep;137:5-17. doi: 10.1016/j.jsbmb.2013.01.007. Epub 2013 Jan 17. J Steroid Biochem Mol Biol. 2013. PMID: 23333934 Review.
Cited by
-
Prenatal Exposure to DEHP Affects Spermatogenesis and Sperm DNA Methylation in a Strain-Dependent Manner.PLoS One. 2015 Aug 5;10(7):e0132136. doi: 10.1371/journal.pone.0132136. eCollection 2015. PLoS One. 2015. PMID: 26244509 Free PMC article.
-
Endocrine-Disrupting Chemicals and Their Adverse Effects on the Endoplasmic Reticulum.Int J Mol Sci. 2022 Jan 29;23(3):1581. doi: 10.3390/ijms23031581. Int J Mol Sci. 2022. PMID: 35163501 Free PMC article. Review.
-
Environmentally-relevant exposure to diethylhexyl phthalate (DEHP) alters regulation of double-strand break formation and crossover designation leading to germline dysfunction in Caenorhabditis elegans.PLoS Genet. 2020 Jan 9;16(1):e1008529. doi: 10.1371/journal.pgen.1008529. eCollection 2020 Jan. PLoS Genet. 2020. PMID: 31917788 Free PMC article.
-
Prenatal DEHP exposure induces lifelong testicular toxicity by continuously interfering with steroidogenic gene expression.Transl Androl Urol. 2024 Mar 31;13(3):369-382. doi: 10.21037/tau-23-503. Epub 2024 Mar 6. Transl Androl Urol. 2024. PMID: 38590960 Free PMC article.
-
Transgenerational Effects of Di-(2-Ethylhexyl) Phthalate (DEHP) on Stress Hormones and Behavior.Endocrinology. 2015 Sep;156(9):3077-83. doi: 10.1210/EN.2015-1326. Epub 2015 Jul 13. Endocrinology. 2015. PMID: 26168342 Free PMC article.
References
-
- Heudorf U, Mersch-Sundermann V, Angerer J. Phthalates: toxicology and exposure. Int J Hyg Environ Health. 2007;210:623–34. - PubMed
-
- Thomas JA, Thomas MJ. Biological effects of di-(2-ethylhexyl) phthalate and other phthalic acid esters. Crit Rev Toxicol. 1984;13:283–317. - PubMed
-
- Latini G. Monitoring phthalate exposure in humans. Clinica Chimica Acta. 2005;361:20–9. - PubMed
-
- Schmitzer JL, Scheunert I, Korte F. Fate of bis(2-ethylhexyl) [14C]phthalate in laboratory and outdoor soil-plant systems. Journal of Agricultural and Food Chemistry. 1988;36:210–5.
-
- Latini G, Verrotti A, De Felice C. DI-2-Ethylhexyl Phthalate and Endocrine Disruption: A Review. Current Drug Targets - Immune, Endocrine & Metabolic Disorders. 2004;4:37–40. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

