Inactivation of Patched1 in mice leads to development of gastrointestinal stromal-like tumors that express Pdgfrα but not kit
- PMID: 23041331
- PMCID: PMC4231777
- DOI: 10.1053/j.gastro.2012.09.061
Inactivation of Patched1 in mice leads to development of gastrointestinal stromal-like tumors that express Pdgfrα but not kit
Abstract
Background & aims: A fraction of gastrointestinal stromal tumor (GIST) cells overexpress the platelet-derived growth factor receptor (PDGFR)A, although most overexpress KIT. It is not known if this is because these receptor tyrosine kinases have complementary oncogenic potential, or because of heterogeneity in the cellular origin of GIST. Little also is known about why Hedgehog (HH) signaling is activated in some GIST. HH binds to and inactivates the receptor protein patched homolog (PTCH).
Methods: Ptch was conditionally inactivated in mice (to achieve constitutive HH signaling) using a Cre recombinase regulated by the lysozyme M promoter. Cre-expressing cells were traced using R26R-LacZ reporter mice. Tumors were characterized by in situ hybridization, immunohistochemistry, immunoblot, and quantitative reverse-transcriptase polymerase chain reaction analyses. Cell transformation was assessed by soft agar assay.
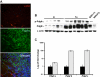
Results: Loss of Ptch from lysozyme M-expressing cells resulted in the development of tumors of GIST-like localization and histology; these were reduced when mice were given imatinib, a drug that targets KIT and PDGFRA. The Hh signaling pathway was activated in the tumor cells, and Pdgfrα, but not Kit, was overexpressed and activated. Lineage tracing revealed that Cre-expressing intestinal cells were Kit-negative. These cells sometimes expressed Pdgfrα and were located near Kit-positive interstitial cells of Cajal. In contrast to KIT, activation of PDGFRA increased anchorage-independent proliferation and was required for tumor formation in mice by cells with activated HH signaling.
Conclusions: Inactivation of Ptch in mice leads to formation of GIST-like tumors that express Pdgfrα, but not Kit. Activation of Pdgfrα signaling appears to facilitate tumorigenesis.
Copyright © 2013 AGA Institute. Published by Elsevier Inc. All rights reserved.
Figures




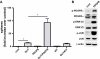
Similar articles
-
Hedgehog pathway dysregulation contributes to the pathogenesis of human gastrointestinal stromal tumors via GLI-mediated activation of KIT expression.Oncotarget. 2016 Nov 29;7(48):78226-78241. doi: 10.18632/oncotarget.12909. Oncotarget. 2016. PMID: 27793025 Free PMC article.
-
Platelet-Derived Growth Factor Receptor-α Regulates Proliferation of Gastrointestinal Stromal Tumor Cells With Mutations in KIT by Stabilizing ETV1.Gastroenterology. 2015 Aug;149(2):420-32.e16. doi: 10.1053/j.gastro.2015.04.006. Epub 2015 Apr 9. Gastroenterology. 2015. PMID: 25865047 Free PMC article.
-
Kitlow stem cells cause resistance to Kit/platelet-derived growth factor alpha inhibitors in murine gastrointestinal stromal tumors.Gastroenterology. 2010 Sep;139(3):942-52. doi: 10.1053/j.gastro.2010.05.083. Epub 2010 Jun 4. Gastroenterology. 2010. PMID: 20621681 Free PMC article.
-
Gastrointestinal stromal tumor and its targeted therapeutics.Chin J Cancer. 2011 May;30(5):303-14. doi: 10.5732/cjc.011.10062. Chin J Cancer. 2011. PMID: 21527063 Free PMC article. Review.
-
The role of KIT in the management of patients with gastrointestinal stromal tumors.Hum Pathol. 2007 May;38(5):679-87. doi: 10.1016/j.humpath.2007.03.001. Hum Pathol. 2007. PMID: 17437861 Review.
Cited by
-
Targeting Disease Persistence in Gastrointestinal Stromal Tumors.Stem Cells Transl Med. 2015 Jul;4(7):702-7. doi: 10.5966/sctm.2014-0298. Epub 2015 May 1. Stem Cells Transl Med. 2015. PMID: 25934947 Free PMC article.
-
Combining hedgehog signaling inhibition with focal irradiation on reduction of pancreatic cancer metastasis.Mol Cancer Ther. 2013 Jun;12(6):1038-48. doi: 10.1158/1535-7163.MCT-12-1030. Epub 2013 Mar 6. Mol Cancer Ther. 2013. PMID: 23468532 Free PMC article.
-
AKT1 Activation is Obligatory for Spontaneous BCC Tumor Growth in a Murine Model that Mimics Some Features of Basal Cell Nevus Syndrome.Cancer Prev Res (Phila). 2016 Oct;9(10):794-802. doi: 10.1158/1940-6207.CAPR-16-0066. Epub 2016 Jul 7. Cancer Prev Res (Phila). 2016. PMID: 27388747 Free PMC article.
-
Hedgehog pathway dysregulation contributes to the pathogenesis of human gastrointestinal stromal tumors via GLI-mediated activation of KIT expression.Oncotarget. 2016 Nov 29;7(48):78226-78241. doi: 10.18632/oncotarget.12909. Oncotarget. 2016. PMID: 27793025 Free PMC article.
-
Targeting hedgehog signaling in cancer: research and clinical developments.Onco Targets Ther. 2013 Oct 10;6:1425-35. doi: 10.2147/OTT.S34678. Onco Targets Ther. 2013. PMID: 24143114 Free PMC article. Review.
References
-
- Nilsson B, Bumming P, Meis-Kindblom JM, et al. Gastrointestinal stromal tumors: the incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate era--a population-based study in western Sweden. Cancer. 2005;103:821–829. - PubMed
-
- Liegl-Atzwanger B, Fletcher JA, Fletcher CD. Gastrointestinal stromal tumors. Virchows Arch. 2010;456:111–127. - PubMed
-
- Sleijfer S, Wiemer E, Seynaeve C, et al. Improved insight into resistance mechanisms to imatinib in gastrointestinal stromal tumors: a basis for novel approaches and individualization of treatment. Oncologist. 2007;12:719–726. - PubMed
-
- Agaram NP, Besmer P, Wong GC, et al. Pathologic and molecular heterogeneity in imatinib-stable or imatinib-responsive gastrointestinal stromal tumors. Clin Cancer Res. 2007;13:170–181. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

