4-O-methylhonokiol, a PPARγ agonist, inhibits prostate tumour growth: p21-mediated suppression of NF-κB activity
- PMID: 23043610
- PMCID: PMC3594673
- DOI: 10.1111/j.1476-5381.2012.02235.x
4-O-methylhonokiol, a PPARγ agonist, inhibits prostate tumour growth: p21-mediated suppression of NF-κB activity
Abstract
Background and purpose: The effects of 4-O-methylhonokiol (MH), a constituent of Magnolia officinalis, were investigated on human prostate cancer cells and its mechanism of action elucidated.
Experimental approach: The anti-cancer effects of MH were examined in prostate cancer and normal cells. The effects were validated in vivo using a mouse xenograft model.
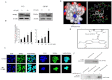
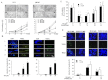
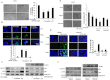
Key results: MH increased the expression of PPARγ in prostate PC-3 and LNCap cells. The pull-down assay and molecular docking study indicated that MH directly binds to PPARγ. MH also increased transcriptional activity of PPARγ but decreased NF-κB activity. MH inhibited the growth of human prostate cancer cells, an effect attenuated by the PPARγ antagonist GW9662. MH induced apoptotic cell death and this was related to G(0) -G(1) phase cell cycle arrest. MH increased the expression of the cell cycle regulator p21, and apoptotic proteins, whereas it decreased phosphorylation of Rb and anti-apoptotic proteins. Transfection of PC3 cells with p21 siRNA or a p21 mutant plasmid on the cyclin D1/ cycline-dependent kinase 4 binding site abolished the effects of MH on cell growth, cell viability and related protein expression. In the animal studies, MH inhibited tumour growth, NF-κB activity and expression of anti-apoptotic proteins, whereas it increased the transcriptional activity and expression of PPARγ, and the expression of apoptotic proteins and p21 in tumour tissues.
Conclusions and implication: MH inhibits growth of human prostate cancer cells through activation of PPARγ, suppression of NF-κB and arrest of the cell cycle. Thus, MH might be a useful tool for treatment of prostate cancer.
© 2012 The Authors. British Journal of Pharmacology © 2012 The British Pharmacological Society.
Figures


Similar articles
-
4-O-methylhonokiol inhibits colon tumor growth via p21-mediated suppression of NF-κB activity.J Nutr Biochem. 2012 Jul;23(7):706-15. doi: 10.1016/j.jnutbio.2011.03.013. Epub 2011 Aug 4. J Nutr Biochem. 2012. PMID: 21820300
-
Honokiol causes G0-G1 phase cell cycle arrest in human prostate cancer cells in association with suppression of retinoblastoma protein level/phosphorylation and inhibition of E2F1 transcriptional activity.Mol Cancer Ther. 2007 Oct;6(10):2686-95. doi: 10.1158/1535-7163.MCT-07-0217. Mol Cancer Ther. 2007. PMID: 17938262
-
Peroxisome proliferator-activated receptor-gamma agonist 4-O-methylhonokiol induces apoptosis by triggering the intrinsic apoptosis pathway and inhibiting the PI3K/Akt survival pathway in SiHa human cervical cancer cells.J Microbiol Biotechnol. 2015 Mar;25(3):334-42. doi: 10.4014/jmb.1411.11073. J Microbiol Biotechnol. 2015. PMID: 25563418
-
The role of peroxisome proliferator-activated receptor gamma in prostate cancer.Asian J Androl. 2018 May-Jun;20(3):238-243. doi: 10.4103/aja.aja_15_17. Asian J Androl. 2018. PMID: 28597850 Free PMC article. Review.
-
Cannabinoids and Prostate Cancer: A Systematic Review of Animal Studies.Int J Mol Sci. 2020 Aug 29;21(17):6265. doi: 10.3390/ijms21176265. Int J Mol Sci. 2020. PMID: 32872551 Free PMC article.
Cited by
-
Targeting miRNAs in renal cell carcinoma: emerging therapeutic strategies.Int J Clin Oncol. 2025 Aug 21. doi: 10.1007/s10147-025-02856-5. Online ahead of print. Int J Clin Oncol. 2025. PMID: 40839233 Review.
-
4-O'-methylhonokiol protects from alcohol/carbon tetrachloride-induced liver injury in mice.J Mol Med (Berl). 2017 Oct;95(10):1077-1089. doi: 10.1007/s00109-017-1556-y. Epub 2017 Jul 8. J Mol Med (Berl). 2017. PMID: 28689299
-
ESS2 controls prostate cancer progression through recruitment of chromodomain helicase DNA binding protein 1.Sci Rep. 2023 Jul 31;13(1):12355. doi: 10.1038/s41598-023-39626-0. Sci Rep. 2023. PMID: 37524814 Free PMC article.
-
4-O-Methylhonokiol Protects HaCaT Cells from TGF-β1-Induced Cell Cycle Arrest by Regulating Canonical and Non-Canonical Pathways of TGF-β Signaling.Biomol Ther (Seoul). 2017 Jul 1;25(4):417-426. doi: 10.4062/biomolther.2016.003. Biomol Ther (Seoul). 2017. PMID: 28190316 Free PMC article.
-
Comparative Study of Docosahexaenoic Acid with Different Molecular Forms for Promoting Apoptosis of the 95D Non-Small-Cell Lung Cancer Cells in a PPARγ-Dependent Manner.Mar Drugs. 2022 Sep 23;20(10):599. doi: 10.3390/md20100599. Mar Drugs. 2022. PMID: 36286423 Free PMC article.
References
-
- Allen CT, Ricker JL, Chen Z, Van Waes C. Role of activated nuclear factor-kappaB in the pathogenesis and therapy of squamous cell carcinoma of the head and neck. Head Neck. 2007;29:959–971. - PubMed
-
- Ban JO, Lee HS, Jeong HS, Song S, Hwang BY, Moon DC, et al. Thiacremonone augments chemotherapeutic agent-induced growth inhibition in human colon cancer cells through inactivation of nuclear factor-{kappa}B. Mol Cancer Res. 2009a;7:870–879. - PubMed
-
- Ban JO, Oh JH, Hwang BY, Moon DC, Jeong HS, Lee S, et al. Inflexinol inhibits colon cancer cell growth through inhibition of nuclear factor-kappaB activity via direct interaction with p50. Mol Cancer Ther. 2009b;8:1613–1624. - PubMed
-
- Biggs JR, Kraft AS. Inhibitors of cyclin-dependent kinase and cancer. J Mol Med (Berl) 1995;73:509–514. - PubMed
-
- Chang PY, Miyamoto S. Nuclear factor-kappaB dimer exchange promotes a p21(waf1/cip1) superinduction response in human T leukemic cells. Mol Cancer Res. 2006;4:101–112. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

