Prophylactic, therapeutic and neutralizing effects of zinc oxide tetrapod structures against herpes simplex virus type-2 infection
- PMID: 23047013
- PMCID: PMC3535478
- DOI: 10.1016/j.antiviral.2012.09.020
Prophylactic, therapeutic and neutralizing effects of zinc oxide tetrapod structures against herpes simplex virus type-2 infection
Abstract
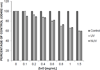
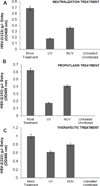
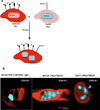
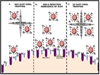
The attachment of Herpes simplex virus type-2 (HSV-2) to a target cell requires ionic interactions between negatively charged cell surface co-receptor heparan sulfate (HS) and positively charged residues on viral envelop glycoproteins, gB and gC. Effective blocking of this first step of HSV-2 pathogenesis demonstrates significant prophylactic effects against the viral disease; any in vitro therapeutic effects of blocking this interaction, however, are not clear. Here, we provide new evidence that zinc oxide tetrapod micro-nanostructures synthesized by flame transport approach significantly block HSV-2 entry into target cells and, in addition, demonstrate the potential to stop the spread of the virus among already infected cells. The zinc oxide tetrapods (ZnOTs) also exhibit the ability to neutralize HSV-2 virions. Natural target cells such as human vaginal epithelial and HeLa cells showed highly reduced infectivity when infected with HSV-2 virions that were pre-incubated with the ZnOTs. The mechanism behind the ability of ZnOTs to prevent, neutralize or reduce HSV-2 infection relies on their ability to bind the HSV-2 virions. We used fluorescently labeled ZnOTs and GFP-expressing HSV-2 virions to demonstrate the binding of the ZnOTs with HSV-2. We also show that the binding and hence, the antiviral effects of ZnOTs can be enhanced by illuminating the ZnOTs with UV light. Our results provide new insights into the anti-HSV-2 effects of ZnOT and rationalize their development as a HSV-2 trapping agent for the prevention and/or treatment of infection. The observed results also demonstrate that blocking HSV-2 attachment can have prophylactic as well as therapeutic applications.
Copyright © 2012 Elsevier B.V. All rights reserved.
Figures








Similar articles
-
Virostatic potential of micro-nano filopodia-like ZnO structures against herpes simplex virus-1.Antiviral Res. 2011 Nov;92(2):305-12. doi: 10.1016/j.antiviral.2011.08.017. Epub 2011 Aug 26. Antiviral Res. 2011. PMID: 21893101 Free PMC article.
-
Anti-heparan sulfate peptides that block herpes simplex virus infection in vivo.J Biol Chem. 2011 Jul 15;286(28):25406-15. doi: 10.1074/jbc.M110.201103. Epub 2011 May 19. J Biol Chem. 2011. PMID: 21596749 Free PMC article.
-
Zinc oxide tetrapods inhibit herpes simplex virus infection of cultured corneas.Mol Vis. 2017 Feb 26;23:26-38. eCollection 2017. Mol Vis. 2017. PMID: 28275313 Free PMC article.
-
Mechanistic Studies of Viral Entry: An Overview of Dendrimer-Based Microbicides As Entry Inhibitors Against Both HIV and HSV-2 Overlapped Infections.Med Res Rev. 2017 Jan;37(1):149-179. doi: 10.1002/med.21405. Epub 2016 Aug 12. Med Res Rev. 2017. PMID: 27518199 Review.
-
Receptors and ligands for herpes simplex viruses: Novel insights for drug targeting.Drug Discov Today. 2022 Jan;27(1):185-195. doi: 10.1016/j.drudis.2021.10.004. Epub 2021 Oct 19. Drug Discov Today. 2022. PMID: 34678489 Review.
Cited by
-
Exploiting herpes simplex virus entry for novel therapeutics.Viruses. 2013 Jun 10;5(6):1447-65. doi: 10.3390/v5061447. Viruses. 2013. PMID: 23752649 Free PMC article. Review.
-
Herpes Simplex Virus: The Hostile Guest That Takes Over Your Home.Front Microbiol. 2020 May 7;11:733. doi: 10.3389/fmicb.2020.00733. eCollection 2020. Front Microbiol. 2020. PMID: 32457704 Free PMC article. Review.
-
Advances in development of antiviral strategies against respiratory syncytial virus.Acta Pharm Sin B. 2025 Apr;15(4):1752-1772. doi: 10.1016/j.apsb.2025.02.015. Epub 2025 Feb 20. Acta Pharm Sin B. 2025. PMID: 40486844 Free PMC article. Review.
-
Zinc Oxide Tetrapods Modulate Wound Healing and Cytokine Release In Vitro-A New Antiproliferative Substance in Glaucoma Filtering Surgery.Life (Basel). 2022 Oct 24;12(11):1691. doi: 10.3390/life12111691. Life (Basel). 2022. PMID: 36362846 Free PMC article.
-
Multi-core-shell composite SnO2NPs@ZIF-8: potential antiviral agent and effective photocatalyst for waste-water treatment.Environ Sci Pollut Res Int. 2019 Aug;26(23):23346-23358. doi: 10.1007/s11356-019-05646-5. Epub 2019 Jun 13. Environ Sci Pollut Res Int. 2019. PMID: 31197665
References
-
- Adelung R, Kaps S, Mishra YK, Claus M, Preusse T, Wolpert C. Germany: 2011.
-
- Baram-Pinto D, Shukla S, Gedanken A, Sarid R. Inhibition of HSV-1 attachment, entry, and cell-to-cell spread by functionalized multivalent gold nanoparticles. Small. 2010;6:1044–1050. - PubMed
-
- Baram-Pinto D, Shukla S, Perkas N, Gedanken A, Sarid R. Inhibition of herpes simplex virus type 1 infection by silver nanoparticles capped with mercaptoethane sulfonate. Bioconjugate chemistry. 2009;20:1497–1502. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

