Prognostic value of 18-fluorodeoxyglucose positron emission tomography-computed tomography in resectable colorectal cancer
- PMID: 23049216
- PMCID: PMC3460334
- DOI: 10.3748/wjg.v18.i36.5072
Prognostic value of 18-fluorodeoxyglucose positron emission tomography-computed tomography in resectable colorectal cancer
Abstract
Aim: To assess the prognostic value of preoperative 18 fluorodeoxyglucose positron emission tomography (FDG-PET)/computed tomography (CT) in patients with resectable colorectal cancer.
Methods: One hundred sixty-three patients with resectable colorectal cancer who underwent FDG-PET/CT before surgery were included. Patient data including pathologic stage at presentation, histology, treatment, disease-free survival and the maximum standardized uptake value (SUVmax) of the primary tumor on FDG-PET/CT were retrospectively analyzed. Median follow up duration was 756 (range, 419-1355). The primary end point was disease-free survival.
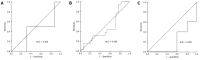
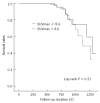
Results: Twenty-five of 163 patients (15.3%) had recurrences. The median SUVmax values of the recurrence and no-recurrence groups were 8.9 (range, 5-24) and 8.2 (range, 0-23, P = 0.998). Receiver operating characteristic (ROC) curve analysis showed no significant association between SUVmax and recurrence (area under the curve = 0.5, P = 0.998, 95% CI: 0.389-0.611). Because a statistically significant value was not found, SUVmax was dichotomized at its median of 8.6. The disease-free survival curve was analyzed using the median SUVmax (8.6) as the cut off. Univariate and multivariate analysis did not provide evidence that disease-free survival rates for the subgroups defined by the median SUVmax were significantly different (P = 0.52, P = 0.25).
Conclusion: Our study suggests that the high FDG uptake of primary mass in resectable colorectal cancer doesn't have a significant relationship with tumor recurrence and disease-free survival.
Keywords: Colorectal neoplasms; Disease-free survival; Positron-emission tomography; Prognosis; Recurrence.
Figures

Similar articles
-
The preoperative SUVmax for (18)F-FDG uptake predicts survival in patients with colorectal cancer.BMC Cancer. 2015 Dec 21;15:991. doi: 10.1186/s12885-015-1991-5. BMC Cancer. 2015. PMID: 26689966 Free PMC article.
-
Incidental focal colorectal ¹⁸F-fluorodeoxyglucose uptake on positron emission tomography/computed tomography.World J Gastroenterol. 2013 Jun 14;19(22):3453-8. doi: 10.3748/wjg.v19.i22.3453. World J Gastroenterol. 2013. PMID: 23801838 Free PMC article.
-
Preoperative 18F-FDG PET/CT predicts disease-free survival in patients with primary invasive ductal breast cancer.Acta Radiol. 2015 Dec;56(12):1463-70. doi: 10.1177/0284185114556929. Epub 2014 Nov 18. Acta Radiol. 2015. PMID: 25406431
-
The prognostic value of SUVmax measuring on primary lesion and ALN by 18F-FDG PET or PET/CT in patients with breast cancer.Eur J Radiol. 2018 Aug;105:1-7. doi: 10.1016/j.ejrad.2018.05.014. Epub 2018 May 17. Eur J Radiol. 2018. PMID: 30017264 Review.
-
Impact of Fluorodeoxyglucose PET/Computed Tomography on the Management of Patients with Colorectal Cancer.PET Clin. 2015 Jul;10(3):345-60. doi: 10.1016/j.cpet.2015.03.007. Epub 2015 Apr 18. PET Clin. 2015. PMID: 26099671 Review.
Cited by
-
Prognostic value of 18F-FDG uptake by regional lymph nodes on pretreatment PET/CT in patients with resectable colorectal cancer.Eur J Nucl Med Mol Imaging. 2014 Dec;41(12):2203-11. doi: 10.1007/s00259-014-2840-5. Epub 2014 Jul 3. Eur J Nucl Med Mol Imaging. 2014. PMID: 24990406
-
Prognostic Impact of Total Lesion Glycolysis (TLG) from Preoperative 18F-FDG PET/CT in Stage II/III Colorectal Adenocarcinoma: Extending the Value of PET/CT for Resectable Disease.Cancers (Basel). 2022 Jan 24;14(3):582. doi: 10.3390/cancers14030582. Cancers (Basel). 2022. PMID: 35158851 Free PMC article.
-
Correlation between long-term aspirin use and F-fluorodeoxyglucose uptake in colorectal cancer measured by PET/CT.PLoS One. 2014 Oct 7;9(10):e109459. doi: 10.1371/journal.pone.0109459. eCollection 2014. PLoS One. 2014. PMID: 25290692 Free PMC article.
-
Preoperative 18F-FDG PET/CT in Patients with Presumed Localized Colon Cancer: A Prospective Study with Long-Term Follow-Up.Cancers (Basel). 2024 Jan 4;16(1):233. doi: 10.3390/cancers16010233. Cancers (Basel). 2024. PMID: 38201660 Free PMC article.
-
Recent chemotherapy reduces the maximum-standardized uptake value of 18F-fluoro-deoxyglucose positron emission tomography in colorectal cancer.Gut Liver. 2014 May;8(3):254-64. doi: 10.5009/gnl.2014.8.3.254. Gut Liver. 2014. PMID: 24827621 Free PMC article.
References
-
- Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. - PubMed
-
- de Geus-Oei LF, Vriens D, van Laarhoven HW, van der Graaf WT, Oyen WJ. Monitoring and predicting response to therapy with 18F-FDG PET in colorectal cancer: a systematic review. J Nucl Med. 2009;50 Suppl 1:43S–54S. - PubMed
-
- Davey K, Heriot AG, Mackay J, Drummond E, Hogg A, Ngan S, Milner AD, Hicks RJ. The impact of 18-fluorodeoxyglucose positron emission tomography-computed tomography on the staging and management of primary rectal cancer. Dis Colon Rectum. 2008;51:997–1003. - PubMed
-
- Lin M, Wong K, Ng WL, Shon IH, Morgan M. Positron emission tomography and colorectal cancer. Crit Rev Oncol Hematol. 2011;77:30–47. - PubMed
-
- Flamen P. Positron emission tomography in colorectal cancer. Best Pract Res Clin Gastroenterol. 2002;16:237–251. - PubMed

