Role of circulating osteogenic progenitor cells in calcific aortic stenosis
- PMID: 23062532
- PMCID: PMC4213791
- DOI: 10.1016/j.jacc.2012.07.042
Role of circulating osteogenic progenitor cells in calcific aortic stenosis
Erratum in
- J Am Coll Cardiol. 2012 Dec 18;60(24):2606. McGregor, Ulrike [added]
Abstract
Objectives: The purpose of this study was to determine the role of circulating endothelial progenitor cells with osteoblastic phenotype (EPC-OCN) in human aortic valve calcification (AVC).
Background: Recent evidence suggests that rather than passive mineralization, AVC is an active atherosclerotic process with an osteoblastic component resembling coronary calcification. We have recently identified circulating EPCs with osteogenic properties carrying both endothelial progenitor (CD34, KDR) and osteoblastic (osteocalcin [OCN]) cell surface markers.
Methods: Blood samples from controls (n = 22) and patients with mild to moderate calcific aortic stenosis (mi-moAS, n = 17), severe calcific AS (sAS, n = 26), and both sAS and severe coronary artery disease (sCAD) (n = 33) were collected during diagnostic coronary angiography. By using flow cytometry, peripheral blood mononuclear cells were analyzed for CD34, KDR, and OCN. Resected normal and calcified aortic valves were analyzed histologically.
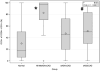
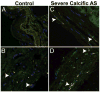
Results: Patients with mi-moAS and patients with sAS/sCAD had significantly less EPCs (CD34+/KDR+/OCN-) than controls. Patients with sAS showed significantly higher numbers of EPC-OCN (CD34+/KDR+/OCN+) than controls. In addition, the percentage of EPC costaining for OCN was higher in all disease groups compared with controls. A subgroup analysis of younger patients with bicuspid sAS showed a similar pattern of significantly lower EPCs but a high percentage of coexpression of OCN. Immunofluorescence showed colocalization of nuclear factor kappa-B and OCN in diseased and normal valves. CD34+/OCN+ cells were abundant in the endothelial and deeper cell layers of calcific aortic valve tissue but not in normal aortic valve tissue.
Conclusions: Circulating EPC-OCN may play a significant role in the pathogenesis and as markers of prognostication of calcific AS.
Copyright © 2012 American College of Cardiology Foundation. Published by Elsevier Inc. All rights reserved.
Figures




Comment in
-
Boning-up on aortic valve calcification.J Am Coll Cardiol. 2012 Nov 6;60(19):1954-5. doi: 10.1016/j.jacc.2012.08.961. Epub 2012 Oct 10. J Am Coll Cardiol. 2012. PMID: 23062540 No abstract available.
References
-
- Lindroos M, Kupari M, Heikkila J, Tilvis R. Prevalence of aortic valve abnormalities in the elderly: an echocardiographic study of a random population sample. J Am Coll Cardiol. 1993;21:1220–5. - PubMed
-
- Nkomo VT, Gardin JM, Skelton TN, Gottdiener JS, Scott CG, Enriquez-Sarano M. Burden of valvular heart diseases: a population-based study. Lancet. 2006;368:1005–11. - PubMed
-
- Ross J, Jr, Braunwald E. Aortic stenosis. Circulation. 1968;38:61–7. - PubMed
-
- Mohler ER, 3rd, Gannon F, Reynolds C, Zimmerman R, Keane MG, Kaplan FS. Bone formation and inflammation in cardiac valves. Circulation. 2001;103:1522–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

