NK cell lytic granules are highly motile at the immunological synapse and require F-actin for post-degranulation persistence
- PMID: 23066148
- PMCID: PMC3558996
- DOI: 10.4049/jimmunol.1201296
NK cell lytic granules are highly motile at the immunological synapse and require F-actin for post-degranulation persistence
Abstract
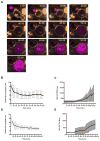
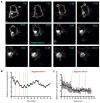
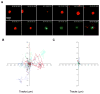
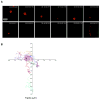
The formation of a dynamic, actin-rich immunological synapse (IS) and the polarization of cytolytic granules toward target cells are essential to the cytotoxic function of NK cells. Following polarization, lytic granules navigate through the pervasive actin network at the IS to degranulate and secrete their toxic contents onto target cells. We examined lytic granule motility and persistence at the cell cortex of activated human NK cells, using high-resolution total internal reflection microscopy and highly quantitative analysis techniques. We illustrate that lytic granules are dynamic and observe substantial motility at the plane of the cell cortex prior to, but not after, degranulation. We also show that there is no significant change in granule motility in the presence of Latrunculin A (which induces actin depolymerization), when added after granule polarization, but that there is a significant decrease in lytic granule persistence subsequent to degranulation. Thus, we show that lytic granules are highly dynamic at the cytolytic human NK cell IS prior to degranulation and that the persistence of granules at the cortex following exocytosis requires the integrity of the synaptic actin network.
Figures




Similar articles
-
Molecular regulation of the plasma membrane-proximal cellular steps involved in NK cell cytolytic function.J Cell Sci. 2020 Feb 21;133(5):jcs240424. doi: 10.1242/jcs.240424. J Cell Sci. 2020. PMID: 32086255 Free PMC article. Review.
-
Nanoscale Dynamism of Actin Enables Secretory Function in Cytolytic Cells.Curr Biol. 2018 Feb 19;28(4):489-502.e9. doi: 10.1016/j.cub.2017.12.044. Epub 2018 Feb 1. Curr Biol. 2018. PMID: 29398219 Free PMC article.
-
UNC-45A Is a Nonmuscle Myosin IIA Chaperone Required for NK Cell Cytotoxicity via Control of Lytic Granule Secretion.J Immunol. 2015 Nov 15;195(10):4760-70. doi: 10.4049/jimmunol.1500979. Epub 2015 Oct 5. J Immunol. 2015. PMID: 26438524 Free PMC article.
-
Remodelling of cortical actin where lytic granules dock at natural killer cell immune synapses revealed by super-resolution microscopy.PLoS Biol. 2011 Sep;9(9):e1001152. doi: 10.1371/journal.pbio.1001152. Epub 2011 Sep 13. PLoS Biol. 2011. PMID: 21931537 Free PMC article.
-
Navigating barriers: the challenge of directed secretion at the natural killer cell lytic immunological synapse.J Clin Immunol. 2010 May;30(3):358-63. doi: 10.1007/s10875-010-9372-y. Epub 2010 Feb 27. J Clin Immunol. 2010. PMID: 20191315 Free PMC article. Review.
Cited by
-
New views of the human NK cell immunological synapse: recent advances enabled by super- and high-resolution imaging techniques.Front Immunol. 2013 Jan 9;3:421. doi: 10.3389/fimmu.2012.00421. eCollection 2012. Front Immunol. 2013. PMID: 23316204 Free PMC article.
-
Bioengineering tools for probing intracellular events in T lymphocytes.WIREs Mech Dis. 2021 Jul;13(4):e1510. doi: 10.1002/wsbm.1510. Epub 2020 Oct 19. WIREs Mech Dis. 2021. PMID: 33073545 Free PMC article. Review.
-
Molecular regulation of the plasma membrane-proximal cellular steps involved in NK cell cytolytic function.J Cell Sci. 2020 Feb 21;133(5):jcs240424. doi: 10.1242/jcs.240424. J Cell Sci. 2020. PMID: 32086255 Free PMC article. Review.
-
Nanoscale Dynamism of Actin Enables Secretory Function in Cytolytic Cells.Curr Biol. 2018 Feb 19;28(4):489-502.e9. doi: 10.1016/j.cub.2017.12.044. Epub 2018 Feb 1. Curr Biol. 2018. PMID: 29398219 Free PMC article.
-
Inflammatory-driven NK cell maturation and its impact on pathology.Front Immunol. 2022 Dec 9;13:1061959. doi: 10.3389/fimmu.2022.1061959. eCollection 2022. Front Immunol. 2022. PMID: 36569860 Free PMC article. Review.
References
-
- Zarcone D, Prasthofer EF, Malavasi F, Pistoia V, LoBuglio AF, Grossi CE. Ultrastructural analysis of human natural killer cell activation. Blood. 1987;69:1725–1736. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

