The blood and vascular cell compatibility of heparin-modified ePTFE vascular grafts
- PMID: 23069711
- PMCID: PMC4065598
- DOI: 10.1016/j.biomaterials.2012.09.046
The blood and vascular cell compatibility of heparin-modified ePTFE vascular grafts
Abstract
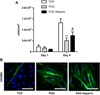
Prosthetic vascular grafts do not mimic the antithrombogenic properties of native blood vessels and therefore have higher rates of complications that involve thrombosis and restenosis. We developed an approach for grafting bioactive heparin, a potent anticoagulant glycosaminoglycan, to the lumen of ePTFE vascular grafts to improve their interactions with blood and vascular cells. Heparin was bound to aminated poly(1,8-octanediol-co-citrate) (POC) via its carboxyl functional groups onto POC-modified ePTFE grafts. The bioactivity and stability of the POC-immobilized heparin (POC-Heparin) were characterized via platelet adhesion and clotting assays. The effects of POC-Heparin on the adhesion, viability and phenotype of primary endothelial cells (EC), blood outgrowth endothelial cells (BOECs) obtained from endothelial progenitor cells (EPCs) isolated from human peripheral blood, and smooth muscle cells were also investigated. POC-Heparin grafts maintained bioactivity under physiologically relevant conditions in vitro for at least one month. Specifically, POC-Heparin-coated ePTFE grafts significantly reduced platelet adhesion and inhibited whole blood clotting kinetics. POC-Heparin supported EC and BOEC adhesion, viability, proliferation, NO production, and expression of endothelial cell-specific markers von Willebrand factor (vWF) and vascular endothelial-cadherin (VE-cadherin). Smooth muscle cells cultured on POC-Heparin showed increased expression of α-actin and decreased cell proliferation. This approach can be easily adapted to modify other blood contacting devices such as stents where antithrombogenicity and improved endothelialization are desirable properties.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures
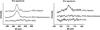








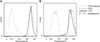
Similar articles
-
Immobilization of heparin/poly-(L)-lysine nanoparticles on dopamine-coated surface to create a heparin density gradient for selective direction of platelet and vascular cells behavior.Acta Biomater. 2014 May;10(5):1940-54. doi: 10.1016/j.actbio.2013.12.013. Epub 2013 Dec 14. Acta Biomater. 2014. PMID: 24342042
-
In vitro and ex vivo hemocompatibility of off-the-shelf modified poly(vinyl alcohol) vascular grafts.Acta Biomater. 2015 Oct;25:97-108. doi: 10.1016/j.actbio.2015.07.039. Epub 2015 Jul 27. Acta Biomater. 2015. PMID: 26225735 Free PMC article.
-
New stent surface materials: the impact of polymer-dependent interactions of human endothelial cells, smooth muscle cells, and platelets.Acta Biomater. 2014 Feb;10(2):688-700. doi: 10.1016/j.actbio.2013.10.015. Epub 2013 Oct 19. Acta Biomater. 2014. PMID: 24148751
-
Strategies for Improving Endothelial Cell Adhesion to Blood-Contacting Medical Devices.Tissue Eng Part B Rev. 2022 Oct;28(5):1067-1092. doi: 10.1089/ten.TEB.2021.0148. Epub 2022 Feb 7. Tissue Eng Part B Rev. 2022. PMID: 34693761 Review.
-
Application of mesenchymal stem cells combined with nano-polypeptide hydrogel in tissue engineering blood vessel.Regen Ther. 2022 Aug 27;21:277-281. doi: 10.1016/j.reth.2022.07.009. eCollection 2022 Dec. Regen Ther. 2022. PMID: 36092503 Free PMC article. Review.
Cited by
-
Glycosaminoglycans: From Vascular Physiology to Tissue Engineering Applications.Front Chem. 2021 May 18;9:680836. doi: 10.3389/fchem.2021.680836. eCollection 2021. Front Chem. 2021. PMID: 34084767 Free PMC article. Review.
-
Sugar Functionalization of Silks with Pathway-Controlled Substitution and Properties.Adv Biol (Weinh). 2021 Jul;5(7):e2100388. doi: 10.1002/adbi.202100388. Epub 2021 Apr 30. Adv Biol (Weinh). 2021. PMID: 33929098 Free PMC article.
-
Advances in the development of tubular structures using extrusion-based 3D cell-printing technology for vascular tissue regenerative applications.Biomater Res. 2022 Dec 5;26(1):73. doi: 10.1186/s40824-022-00321-2. Biomater Res. 2022. PMID: 36471437 Free PMC article. Review.
-
Insights into Adsorption Characterization of Sulfated Xylans onto Poly(ethylene terephthalate).Polymers (Basel). 2020 Apr 5;12(4):825. doi: 10.3390/polym12040825. Polymers (Basel). 2020. PMID: 32260479 Free PMC article.
-
Biodegradable scaffold with built-in vasculature for organ-on-a-chip engineering and direct surgical anastomosis.Nat Mater. 2016 Jun;15(6):669-78. doi: 10.1038/nmat4570. Epub 2016 Mar 7. Nat Mater. 2016. PMID: 26950595 Free PMC article.
References
-
- Curi MA, Skelly CL, Meyerson SL, Woo DH, Desai TR, McKinsey JF, et al. Conduit choice for above-knee femoropopliteal bypass grafting in patients with limb-threatening ischemia. Ann Vasc Surg. 2002;16(1):95–101. - PubMed
-
- Albers M, Battistella VM, Romiti M, Rodrigues AA, Pereira CA. Meta-analysis of polytetrafluoroethylene bypass grafts to infrapopliteal arteries. J Vasc Surg. 2003;37(6):1263–1269. - PubMed
-
- Lord MS, Yu W, Cheng B, Simmons A, Poole-Warren L, Whitelock JM. The modulation of platelet and endothelial cell adhesion to vascular graft materials by perlecan. Biomaterials. 2009;30(28):4898–4906. - PubMed
-
- Wissink MJ, Beernink R, Pieper JS, Poot AA, Engbers GH, Beugeling T, et al. Immobilization of heparin to EDC/NHS-crosslinked collagen. Characterization and in vitro evaluation. Biomaterials. 2001;22(2):151–163. - PubMed
-
- Chandy T, Das GS, Wilson RF, Rao GH. Use of plasma glow for surface-engineering biomolecules to enhance blood compatibility of Dacron and PTFE vascular prosthesis. Biomaterials. 2000;21(7):699–712. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

